
American
Patients First
The Trump Administration Blueprint to Lower Drug Prices
and Reduce Out-of-Pocket Costs
MAY
The U.S. Department of Health & Human Services
Hubert H. Humphrey Building
200 Independence Avenue, S.W.
Washington, D.C. 20201
Toll Free Call Center: 1-877-696-6775

American
Patients First
The Trump Administration Blueprint to Lower Drug Prices
and Reduce Out-of-Pocket Costs
MAY


One of my greatest priorities is to reduce
the price of prescription drugs. In
many other countries, these drugs cost
far less than what we pay in the United
States. That is why I have directed
my Administration to make fixing the
injustice of high drug prices one of our
top priorities. Prices will come down.”
— PRESIDENT DONALD J. TRUMP
“

| 4 | American Patients First

| 5 |
T S H H S
W, D.C.
Message from the Secretary
THE UNITED STATES is first in the world
in biopharmaceutical investment
and innovation. Combining our free
market system and generous pub-
lic investment made America home
to the first chemotherapy treat-
ments for cancer, the first effective
treatments for HIV, the first cure
for Hepatitis C, and now, the first
therapies that turn our own immune
systems against cancer.
But too often, this system has not
put American patients first. We have
access to the greatest medicines in
the world, but access is meaningless
without affordability.
When it comes to the cost of pre-
scription drugs, our healthcare sys-
tem faces four major challenges: high
list prices for drugs; seniors and gov-
ernment programs overpaying for
drugs due to lack of the latest nego-
tiation tools; high and rising out-of-
pocket costs for consumers; and for-
eign governments free-riding off of
American investment in innovation.
Alex M. Azar II
These problems have often been
discussed, but gone unaddressed.
Under President Trump, that has now
changed. This blueprint is a historic
plan for bringing down the high price
of drugs and reducing out-of-pocket
costs for the American consumer.
The time to act is now: Not only
are costs spiraling out of control, but
the scientific landscape is changing
as well. Securing the next generation
of cures for the next generation of
American patients will require radi-
cal reforms to how our system works.
Our blueprint will bring immediate
relief to American patients while also
delivering long-term reforms.
The men and women of the
Department of Health and Human
Services (HHS) are looking at every
facet of HHS’s programs, author-
ities, and spending. Working with
our partners in the private sector,
we will turn this vision into action,
and thereby improve the health and
well-being of every American.

CONTENTS
I. Trump Administration Blueprint in Brief............................................................9
II. What’s the Problem? .............................................................................................. 12
III. Trump Administration Accomplishments on Drug Pricing ........................18
IV. Responding to President Trump’s Call to Action .......................................... 23
V. Further Actions Under Review and Opportunities for Feedback .................26

| 8 | American Patients First

I. Trump Administration Blueprint In Brief | 9 |
I. Trump Administration
Blueprint in Brief
HHS has identified four challenges in the
American drug market:
• High list prices for drugs
• Seniors and government programs overpaying for
drugs due to lack of the latest negotiation tools
• High and rising out-of-pocket costs for consumers
• Foreign governments free-riding o of American
investment in innovation
Under President Trump, HHS has proposed a comprehen-
sive blueprint for addressing these challenges, identify-
ing four key strategies for reform:
• Improved competition
• Better negotiation
• Incentives for lower
list prices
• Lowering out-of-pocket costs
HHS’s blueprint encompasses two phases: 1) actions the
President may direct HHS to take immediately and 2)
actions HHS is actively considering, on which feedback is
being solicited.
| 10 | American Patients First
Increased Competition
Immediate Actions
• Steps to prevent manufacturer
gaming of regulatory processes
such as Risk Evaluation and
Mitigation Strategies (REMS)
• Measures to promote innovation
and competition for biologics
• Developing proposals to stop
Medicaid and Aordable Care
Act programs from raising prices
in the private market
Further Opportunities
• Considering how to
encourage sharing of samples
needed for generic drug
development
• Additional eorts to promote
the use of biosimilars
Better Negotiation
Immediate Actions
• Experimenting with
value-based purchasing
in federal programs
• Allowing more substitution
in Medicare Part D to address
price increases for single-
source generics
• Reforming Medicare Part D to
give plan sponsors significantly
more power when negotiating
with manufacturers
• Sending a report to the President
on whether lower prices on some
Medicare Part B drugs could be
negotiated for by Part D plans
• Leveraging the Competitive
Acquisition Program in Part B.
• Working across the
Administration to assess the
problem of foreign free-riding
Further Opportunities
• Considering further use of
value-based purchasing in
federal programs, including
indication-based pricing and
long-term financing
• Removing government
impediments to value-based
purchasing by private payers
• Requiring site neutrality
in payment
• Evaluating the accuracy
and usefulness of current
national drug spending data
I. Trump Administration Blueprint In Brief | 11 |
Incentives for Lower
List Prices
Immediate Actions
• FDA evaluation of requiring
manufacturers to include list
prices in advertising
• Updating Medicare’s drug-pricing
dashboard to make price increases
and generic competition
more transparent
Further Opportunities
• Measures to restrict the use of
rebates, including revisiting
the safe harbor under the Anti-
Kickback statute for drug rebates
• Additional reforms to the
rebating system
• Using incentives to discourage
manufacturer price increases for
drugs used in Part B and Part D
• Considering fiduciary status
for Pharmacy Benefit
Managers (PBMs)
• Reforms to the Medicaid Drug
Rebate Program
• Reforms to the 340B drug
discount program
• Considering changes to
HHS regulations regarding
drug copay discount cards
Lowering Out-of-Pocket Costs
Immediate Actions
• Prohibiting Part D contracts
from preventing pharmacists
telling patients when they
could pay less out-of-pocket
by not using insurance
• Improving the usefulness
of the Part D Explanation
of Benefits statement by
including information about
drug price increases and
lower cost alternatives
Further Opportunities
• More measures to inform
Medicare Parts B and D
beneficiaries about lower-
cost alternatives
• Providing better annual, or more
frequent, information on costs to
Part D beneficiaries

Insurance
Contract
Reimbursement for Consumers’ Rx
Share of Manufacturer Rebates
Consumers
Payers
Drug
Manufacturer
Pharmacies
Pharmacy
Benefits Manager
Formulary Agreement
Copayment
Network Agreement
PBM Agreement
Payment for
Dispensed Drugs
Formulary
Rebates & Other Fees
Premium
Drugs
Money
Contracting
Dispensed
Drugs
Prime Vendor Agreement
Shipped Bulk Drugs
Payment for
Wholesale Drugs
Distributor
Payment for
Wholesale Drugs
Shipped Bulk Drugs
Distributor Agreement
| 12 | American Patients First
II. What’s the Problem?
TODAY’S COMPLEX U.S. PHARMACEUTI-
CAL MARKET is characterized by high
and rising list prices, increasing
consumer out-of-pocket costs, and
a new era of high-cost drugs lacking
competition. Recent developments
and challenges in the market include
a business model built on opaque re-
bates and discounts that favor high
list prices, a generational loss of
patent exclusivity, the Affordable
Care Act’s taxes, rebates, and ex-
pansion of the 340B drug discount
program, expansion of internation-
al price controls, government pro-
grams lacking modern negotiation
tools, and changes in insurance ben-
efit design that shifted the burden of
rising prices to consumers.
i. A Recent Drug Pricing History
A Business Model Is Born
Thirty years ago, the majority of pre-
scriptions filled at retail pharmacies
were cash transactions. Over time,
however, health plans began to oer
drug coverage to compete for new
members, knowing the benefit could
be oered at relatively low cost.
But the complexity, price, and
benefits associated with prescrip-
tion drugs dramatically increased
during the 1990s. U.S. drug spend-
ing grew between 11 and 17% per
year in the 1990s, as both prices and
volume soared.
1
As more complex and more ex-
pensive drugs came on the market,
FIGURE 1
ADAPTED FROM: Fein,
Adam. J., The 2016
Economic Report on
Retail, Mail and Specialty
Pharmacies, Drug Channels
Institute, January 2016.
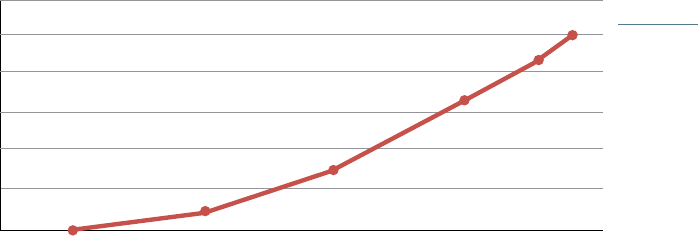
BILLIONS
$
$
$
$
$
$
$
$
$-
$ GROWTH
Ii. What’s The Problem | 13 |
FIGURE 2
Retail Prescription
Drug Spend
SOURCE
CMS Oice of the Actuary
plans used formularies and copay-
ments to manage utilization and
keep drug costs low. But drug manu-
facturers paid rebates and discounts
to be oered as preferred drugs with
lower copays, and few drugs were
excluded from coverage.
In response to rising prices, private
health plans began to use closed for-
mularies to manage drug spending and
negotiate higher rebates and discounts
from drug manufacturers, holding
down increases in net drug prices.
Medicare and Medicaid Evolve
Medicare Part D introduced new dy-
namics to the market, as the first
truly insured prescription drug ben-
efit. Part D plan sponsors agreed to
accept the financial risk of providing
covered drugs to Medicare benefi-
ciaries in exchange for a per-benefi-
ciary, per-month payment.
Part D Plans negotiated ag-
gressively to keep premiums and
drug costs low for cost-conscious
Medicare beneficiaries, and the Part
D Plan Finder brought transparency
to premiums, formularies, and drug
prices. Private health plans outside
of Medicare adopted the tools used
by Part D plans, and Part D has in
large part succeeded at holding down
costs for seniors.
Meanwhile, drug spending has
been held down in the Medicaid pro-
gram by other tools. The program’s
rules prohibit the use of closed for-
mularies, but states use “preferred
drug lists” to negotiate larger sup-
plemental rebates than required by
law and limit the use of drugs made
by manufacturers not oering sup-
plemental rebates. As a result, drug
manufacturers have faced pressure
to oer higher rebates to main-
tain volume, or risk losing revenue
growth caused from being excluded
from markets.
A Generational Loss of Exclusivity
Around two decades after the boom
in drug spending of the 1990s, be-
ginning in 2012, the expiration of
patents of popular drugs—many
“blockbuster drugs” with U.S. sales
| 14 | American Patients First
of $1 billion or more—led to the loss
of over $140 billion in drug manu-
facturer revenue.
2
New generic com-
petition coincided with a slowdown
in new product development, creat-
ing additional financial pressure.
Aordable Care Act Taxes
and Rebates
The Affordable Care Act (ACA)
shifted costs and changed the
Medicaid Drug Rebate Program in
ways that may have driven up pric-
es for consumers, especially in the
private market.
The ACA also created a new tax
on branded prescription drug sales
to Medicare, Medicaid, and other
government health care programs.
Drug companies paid $2.5 billion in
2011, based on their market share
in government programs, a number
that increased to $4.1 billion in 2018.
The ACA also increased the manda-
tory Medicaid base rebate on brand
name drugs to 23.1%, and extended
the Medicaid rebate to drugs pur-
chased by Medicaid Managed Care
Organizations, more than doubling
the number of Medicaid covered
lives using rebate-eligible drugs.
3,4
This expansion of discounts may
have placed pressure on list prices by
forcing drug manufacturers to raise
prices overall.
340B Growth
The ACA also increased the demands
on the 340B drug discount program:
For one, it made critical access hos-
pitals and other hospital types el-
igible for the first time, while the
ACA’s Medicaid expansion made
more hospitals eligible by increas-
ing their Disproportionate Share
Hospital enrollment.
5
In fact, the number of 340B hos-
pitals grew from nearly 1,700 in 2011
to 2,479 in 2017. The number of
non-hospital covered entities, o-
site clinics or “child sites,” hospital
outpatient departments, and con-
tract pharmacies also grew substan-
tially.
6
As a result, discounted drug
purchases made by covered entities
under the 340B program totaled
more than $16 billion in 2016—
nearly a 400% increase in purchases
from 2009.
7
The additional billions
of dollars in discounted sales and the
cross-subsidization necessary may
have created additional pressure on
manufacturers to increase list price.
Growth in International
Price Controls
The global financial crisis in 2008
spurred austerity measures in
most European countries, includ-
ing more aggressive use of existing
drug price controls. Between 2010
and 2011, 23 countries implement-
ed 89 distinct measures to contain
government spending on prescrip-
tion drugs. Most used their sin-
gle-payer healthcare systems to
impose drug price controls along-
side increased copayments, val-
ue-added tax rates on prescription
drugs, and other measures.
8
In 2013, the World Health
Organization published a paper de-

Ii. What’s The Problem | 15 |
scribing the growing use of exter-
nal reference pricing, or the prac-
tice of using the price of a medicine
in one or several countries to derive
a benchmark or reference price for
the purposes of setting or negotiat-
ing the price of the product in a giv-
en country.
9
Every time one country
demands a lower price, it leads to a
lower reference price used by oth-
er countries. Such price controls,
combined with the threat of market
lockout or intellectual property in-
fringement, prevent drug compa-
nies from charging market rates for
their products, while delaying the
availability of new cures to patients
living in countries implementing
these policies.
ii. Where We Are Today
Industry Impact
The loss of patent exclusivity on suc-
cessful products, new ACA taxes, and
requirements to extend higher re-
bates and discounts to a markedly in-
creased Medicaid and 340B popula-
tion created an estimated $200 billion
of downward pressure on pharma-
ceutical industry revenues—during
a five-year period when innovation
was decreasing. International price
controls and delayed global product
launches exacerbated the problem.
Absent new products to launch
and the ability to increase revenue
through volume, and in the face of
a more sophisticated PBM industry
demanding higher rebates and re-
stricting access to markets, the in-
One in seven
employees, and
a growing number
of people with
individually-
purchased
insurance, now
have a separate
drug deductible.
dustry turned to its remaining tool
to drive growth: increasing price.
Prices soared on certain advanced
small molecule drugs and new spe-
cialty drugs. Meanwhile, PBMs ex-
ploited new utilization management
tools and “price protection” con-
tracts to extract even higher rebates,
further widening the gap between
list and net prices.
Each increase in list prices sat-
isfied the drug industry’s need to
grow revenue and increased admin-
istrative fees paid to PBMs, but also
boosted the prices paid by payers
and, especially, consumers.
Lack of Modern Negotiation Tools
in Government Programs
Private health plans in non-govern-

FIGURE 3
List Price vs.
Net Price
SOURCE
Medicine Use and Spending in
the U.S.; A Review of 2017 and
Outlook to 2022. April 19, 2018
Retai Net
Retai Gross
330
310
290
270
250
Biions ($)
230
210
| 16 | American Patients First
ment programs use aggressive formu-
lary management tools to negotiate
better deals for high-cost drugs dis-
pensed in pharmacies. Private health
plans are also presently experimenting
with competitive acquisition and utili-
zation management of drugs adminis-
tered in physicians’ oces.
Medicare rules limit the ability of
Part D plans to use these tools, mak-
ing it harder for plans to negotiate as
eectively for drugs, particular high-
cost drugs that lack competition.
Changes in Beneft Design
Consumers are more and more being
exposed to the rising cost of their
prescription drugs.
In 2016, nearly 40% of adults with
employer-sponsored insurance, and
over half of adults with individual-
ly purchased insurance, enrolled in a
high-deductible
health plan. One in
seven employ
ees, and a growing num-
ber of people with individually-pur-
chased insurance, now have a sepa-
rate drug deductible.
10
Private health
plans in both the employer-sponsored
and individually-purchased markets
are more frequently relying on pre-
scription drug coinsurance regardless
of formulary tier, although they do so
most often for non-preferred brands
and specialty drugs.
Consumers who have not met
their deductible or are subject to
coinsurance, pay based on the
pharmacy list price, which is not
reduced by the substantial drug
manufacturer rebates paid to PBMs
and health plans. As a result, the
growth in list prices, and the wid-
ening gap between list and net
prices, markedly increases con-
sumer out-of-pocket spending,
particularly for high-cost drugs
not subject to negotiation.
This is not only a financial chal-
lenge, but a health issue as well: One
study found that consumers asked
to pay $50 or more at the pharma-
cy counter are four times more likely
to abandon the prescription than a
consumer charged $10.
11

Speciaty Spend
Traditiona Spend
Biions ($)
700
600
500
400
300
200
100
-
% Decrease
% Increase
Ii. What’s The Problem | 17 |
What had been a hidden negotia-
tion and wealth transfer between drug
manufacturers and PBMs is now a
direct increase on consumer out-of-
pocket spending that likely decreases
drug adherence and health outcomes.
The Growth of High-Cost Drugs
New challenges are emerging onto
this landscape. A growing number of
complex drugs account for a grow-
ing percentage of health care spend-
ing. The pharmaceutical industry
has shifted its attention to high-cost
drugs that face little to no competi-
tion, because they oer the freedom
to set high launch prices and in-
crease them over time.
Though these drugs oer hope
to the 1 percent of insured benefi-
ciaries who use them, they account
for 35-40% of health plan spending,
and will increase to over half of drug
spending over the next 5 years.
12
Absent reform, the growth of high
cost drugs will further compound
the issues already described.
FIGURE 4
Growth in
Specialty Drug
Spending
SOURCE
Medicine Use and Spending in
the U.S.; A Review of 2017 and
Outlook to 2022. April 19, 2018
The Situation Today
Taken together, these drivers con-
tribute to high and rising list prices
and consumer out-of-pocket spend-
ing. Because health plans, pharma-
cy benefit managers (PBMs), and
wholesalers receive higher rebates
and fees when list prices increase,
there is little incentive to control
list prices. Consumers, however, pay
higher copayments, coinsurance, or
pre-deductible out-of-pocket costs
when list prices rise.
The Trump Administration be-
lieves it is time to realign the sys-
tem in four ways: increasing com-
petition, improving government
negotiation tools, creating in-
centives for lower list prices, and
bringing down out-of-pocket costs
for consumers.
| 18 | American Patients First
III. Trump Administration
Accomplishments on Drug Pricing
THE PRESIDENT has consistently em-
phasized the need to reduce the price
of prescription drugs. The Trump
Administration has already taken
a number of significant adminis-
trative steps, and proposed in the
President’s FY2019 Budget, to im-
prove competition and end the gam-
ing of regulatory processes, support
better negotiation of drug discounts
through government insurance pro-
grams, create incentives for phar-
maceutical companies to lower list
prices, and reduce consumer out-
of-pocket spending at the pharmacy
and other care settings.
A. Increasing Competition
Since the beginning of the Trump
Administration, HHS has taken a
number of actions to increase com-
petition and end the gaming of regu-
latory processes that may keep drug
prices artificially inflated or hin-
der generic, branded, or biosimilar
competition. These eorts include:
• Accelerating Food and Drug
Administration (FDA) approval of
generic drugs. Studies show that
greater generic competition is
associated with lower prices. FDA
is publishing the names of drugs
that have no competitors in order
to spur new entrants and bring
prices down. Over 1,000 generic
drugs were approved in 2017,
which is the most in FDA’s history
in a calendar year by over 200
drugs. These generic approvals
saved American consumers and
taxpayers nearly $9 billion in 2017.
• Drug Competition Action
Plan. In 2017, President
Trump’s FDA established a
Drug Competition Action Plan
to enable patients to access
more affordable medications
by focusing the agency’s
efforts in three key areas: (1)
improving the efficiency of
the generic drug development,
review, and approval process;
(2) maximizing scientific and
regulatory clarity with respect
to complex generic drugs; and
(3) closing loopholes that allow
brand-name drug companies
to “game” FDA rules in ways
that forestall the generic
competition Congress intended.
The agency also has taken
steps to prioritize its review
of generic drug applications;
issued guidance to improve
efficiencies in the development,
review, and approval processes
for generic drugs, including
Iii. Trump Administration Accomplishments On Drug Pricing | 19 |
complex generic drugs; and
issued guidance to further
streamline the submission
and review process for shared
system Risk Evaluation and
Mitigation Strategies (REMS),
and to allow collective
submissions to streamline
the review of shared REMS.
• FDA also announced it will
facilitate opportunities for
enhanced information sharing
between manufacturers, doctors,
patients and insurers to improve
patient access to medical
products, including through
value-based insurance.
• Speeding access to more
aordable generics by spurring
competition. Today, a generic
manufacturer that has been
awarded 180-day exclusivity for
being the first generic to file can
“park” its application with FDA,
preventing additional generic
manufacturers from entering the
market. The President’s FY2019
Budget proposes to prevent
companies from using their 180-
day exclusivity to indefinitely
delay real competition and
savings for consumers by seeking
a legislative change to start a
company’s 180-day exclusivity
clock in certain instances when
another generic application is
ready for approval, but is blocked
solely by such a first applicant’s
180-day exclusivity.
• Finalizing a policy in which each
biosimilar for a given biologic
gets its own billing and payment
code under Medicare Part B,
to incentivize development of
additional lower-cost biosimilars.
Prior approaches to biosimilar
coding and payment would have
created a race to the bottom of
biosimilar pricing, while leaving
the branded product untouched,
making it an unviable market that
few would want to enter.
B. Better Negotiation
Medicare Part D has been very suc-
cessful since it launched in 2006.
However, prescription drug markets
are dierent than they were 12 years
ago, and in some cases Part D plan
sponsors may be prohibited from
doing what private payers outside
the Medicare program do to nego-
tiate eectively and keep costs low.
More can also be done across the
Medicare program to provide ben-
eficiaries with the lower costs and
greater price transparency resulting
from better negotiation.
Since the beginning of the Trump
Administration, HHS has taken a
number of actions to support better
negotiation. These eorts include:
• Finalizing changes to the
Medicare Prescription Drug
Program in the 2019 Part C and
Part D regulation allowing for
faster mid-year substitution of
generic drugs onto formularies.

| 20 | American Patients First
“We’re the largest
buyer of drugs in
the world, and
yet we d
on’t bid
properly.”
–
PRESIDENT TRUMP
• Proposing in the President’s
FY2019 Budget
i
a 5-part plan to
modernize the Medicare Part
D program, a portion of which
includes enhancing Part D
plans’ negotiating power with
manufacturers by changing Part
D plan formulary standards to
require a minimum of one drug
per category or class rather than
two. We note that the 5-part plan
is intended to be implemented
together, as eliminating even one
piece of the package significantly
changes the proposal’s impacts.
• Proposing in the President’s
FY2019 Budget to address abusive
drug pricing by manufacturers
by: establishing an inflation limit
for reimbursement of Medicare
Part B drugs; reducing Wholesale
Acquisition Cost (WAC)-Based
Payment when Average Sales
Price (ASP) isn’t available; and
improving manufacturers’
reporting of Average Sales Prices
to set accurate payment rates.
• Increasing the integrity of the
Medicaid Drug Rebate Program,
so that manufacturers pay their
fair share in rebates, by proposing
in the President’s FY2019 Budget
to remove ambiguity regarding
how drugs should be reported
under the program. HHS is also
manually reviewing each new
drug that has been reported in
the Medicaid rebate system on
a quarterly basis to make sure
classifications are correct, and
the United States took legal
action against Mylan for its
misclassification of EpiPen,
resulting in an agreement for
Mylan to pay back $465 million in
rebate payments.
• Proposing in the President’s
FY2019 Budget to further clarify
the Medicaid definition of brand
drugs, which would address
inappropriate interpretations
leading some manufacturers to
classify certain brand and over-
the-counter drugs as generics
for Medicaid rebate purposes,
reducing the rebates they owe.
• Proposing in the President’s
FY2019 Budget to call for new
Medicaid demonstration authority
i. https://www.whitehouse.gov/wp-content/uploads/2018/02/budget-fy2019.pdf
Iii. Trump Administration Accomplishments On Drug Pricing | 21 |
for up to five states to test drug
coverage and financing reforms
that build on private sector best
practices. Participating states
would determine their own
drug formularies, coupled with
an appeals process to protect
beneficiary access to non-
covered drugs based on medical
need, and negotiate drug prices
directly with manufacturers.
HHS and participating states
would rigorously evaluate these
demonstrations, which would
provide states with new tools to
control drug costs and tailor drug
coverage decisions to state needs.
• Proposing in the President’s
FY2019 Budget to authorize
the HHS Secretary to leverage
Medicare Part D plans’
negotiating power for certain
drugs covered under Part B.
• Addressing price disparities in
the international market. The
Administration is updating a
number of historical studies
to analyze drug prices paid
in countries that are a part
of the Organisation for
Economic Co-operation and
Development (OECD).
C. Creating Incentives
to Lower List Prices
The list price of a drug does not re-
flect the discounts or price conces-
sions paid to a PBM, insurer, health
plan, or government program.
Obscuring these discounts can shift
costs to consumers in commercial
health plans and Medicare bene-
ficiaries. Many incentives in the
current system reward higher list
prices, and HHS is interested in cre-
ating new incentives to reward drug
manufacturers that lower list prices
or do not increase them.
Since the beginning of the Trump
Administration, HHS has taken a
number of actions to create incen-
tives to lower list prices.
These eorts include propos-
ing in the President’s FY2019 bud-
get a 5-part plan to modernize the
Medicare Part D program, a portion
of which includes the exclusion of
manufacturer discounts from the
calculation of beneficiary out-of-
pocket costs in the Medicare Part
D coverage gap, and the establish-
ment of a beneficiary out-of-pock-
et maximum in the Medicare Part D
catastrophic phase to reduce out-
of-pocket spending for beneficia-
ries who spend the most on drugs.
The changes in the catastrophic
phase would shift more responsibil-
ity onto plans, creating incentives
for plans to negotiate with manu-
facturers to lower prices for high-
cost drugs. We note that the 5-part
plan is intended to be implemented
together, as eliminating even one
piece of the package significantly
changes the proposal’s impacts.
In addition, the President’s
FY2019 Budget proposes reforms to
improve 340B Program integrity and
| 22 | American Patients First
ensure that the benefits derived from
participation in the program are used
to benefit patients, especially low-in-
come and uninsured populations.
D. Reducing Patient
Out-of-Pocket Spending
American patients have the right to
know what their prescription drugs
will really cost before they get to the
pharmacy or get the drug. Too many
people abandon their prescriptions
at the pharmacy when they discover
the price is too high, and too many
patients are never informed of lower
cost options.
Since the beginning of the Trump
Administration, HHS has taken a
number of steps to lower consumer
out-of-pocket spending and improve
transparency. These eorts include:
• Finalizing Medicare Outpatient
Prospective Payment System (OPPS)
rules to reduce beneficiary out-of-
pocket spending for 340B drugs
administered in certain hospitals by
an estimated $320 million in 2018,
which would equal $3.2 billion when
multiplied over ten years.
• Seeking information about
changes in the Medicare
Prescription Drug Program
regulations for contract year 2019
that would increase transparency
for people with Medicare
prescription drug coverage.
The proposed rule included a
Request for Information soliciting
comment on potential policy
approaches for applying some
manufacturer rebates and all
pharmacy price concessions to the
price of a drug at the point of sale.
• Finalizing changes to the
Medicare Prescription Drug
Program in the 2019 Part C
and Part D regulation allowing
Medicare beneficiaries receiving
low-income subsidies to access
biosimilars at a lower cost.
• Proposing in the President’s
FY2019 Budget a 5-part plan to
modernize the Medicare Part
D program, a portion of which
includes eliminating cost-
sharing on generic drugs for
low-income beneficiaries and
requiring Medicare Part D plans
to apply a substantial portion
of rebates at the point of sale.
We note that the 5-part plan
is intended to be implemented
together, as eliminating even one
piece of the package significantly
changes the proposal’s impacts.
We also note that in the months
following this Part D proposed
rule and the President’s budget
proposal that included this policy
change explicitly, several major
insurers and pharmacy benefit
managers announced they would
pass along a portion of rebates
to individual members in their
fully-insured populations
or when otherwise requested
by employers.
Iv. Responding To President Trump’s Call To Action | 23 |
IV. Responding to President
Trump’s Call to Action
PRESIDENT TRUMP has called on the
Administration to propose new
strategies and take bold actions to
improve competition and end the
gaming of regulatory processes,
support better negotiation of drug
discounts through government in-
surance programs, create incentives
for pharmaceutical companies to
lower list prices, and reduce con-
sumer out-of-pocket spending at
the pharmacy and other care set-
tings. HHS may undertake these and
other actions, to the extent permit-
ted by law, in response to President
Trump’s call to action.
A. Improve Competition
In response to President Trump’s
call to action, HHS may support im-
proved competition by:
• Taking steps to prevent gaming
of regulatory processes: FDA
will issue guidance to address
some of the ways in which
manufacturers may seek to use
shared system REMS to delay or
block competition from generic
products entering the market.
• Promoting innovation and
competition for biologics. FDA
will issue new policies to improve
the availability, competitiveness,
and adoption of biosimilars as
aordable alternatives to branded
biologics. FDA will also continue
to educate clinicians, patients,
and payors about biosimilar and
interchangeable products as we
seek to increase awareness about
these important new treatments.
B. Better Negotiation
In response to President Trump’s
call to action, HHS may support bet-
ter negotiation by:
• Directing Centers for Medicare
& Medicaid Services (CMS) to
develop demonstration projects to
test innovative ways to encourage
value-based care and lower drug
prices. These models should
hold manufacturers accountable
for outcomes, align with CMS
priorities of value over volume
and site-neutral payments, and
provide Medicare providers,
payers, and states with additional
tools to manage spending for
high-cost therapies.
• Allowing Part D plans to adjust
formulary or benefit design
during the benefit year if
necessary to address a price
| 24 | American Patients First
increase for a sole source generic
drug. Presently, Part D plans
do not contract with generic
drug manufacturers for the
purchase of generic drugs, and
generally are not permitted
to change their formulary or
benefit design without CMS
approval in response to a price
increase. This change could
ensure Part D plans can respond
to a price increase by the only
manufacturer of a generic drug.
• Providing plans full flexibility
to manage high-cost drugs
that do not provide Part D plans
with rebates or negotiated
fixed prices, including in the
protected classes. Presently,
Part D plans are unable to
negotiate lower prices for high-
cost drugs without competition.
This change could allow Part D
plans to use the tools available
to private payers outside of the
Medicare program to better
negotiate for these drugs.
• Updating the methodology used
to calculate Drug Plan Customer
Service star ratings for plans
that are appropriately managing
utilization of high-cost drugs.
Presently, if a Part D plan issues
an adverse redetermination
decision, the enrollee, the
enrollee’s representative or the
enrollee’s prescriber may appeal
the decision to the Independent
Review Entity (IRE). This process
may discourage Part D plan
sponsors from appropriately
managing utilization of high-
cost drugs. This change could
provide Part D plan sponsors
with the ability to appropriately
manage high-cost changes, while
holding sponsors accountable
primarily using other successful
enforcement mechanisms.
• Evaluating options to allow
high-cost drugs to be priced
or covered dierently based on
their indication. Presently, Part
D plans must cover and pay the
same price for a drug regardless
of the indication for which it was
prescribed. This change could
permit Part D plans to choose to
cover or pay a dierent price for a
drug, based on the indication.
• Sending the President a report
identifying particular drugs or
classes of drugs in Part B where
there are savings to be gained by
moving them to Part D.
• Taking steps to leverage
the authority created by the
Competitive Acquisition
Program (CAP) for Part B Drugs
& Biologicals. This program will
generally provide physicians a
choice between obtaining these
drugs from vendors selected
through a competitive bidding
process or directly purchasing
these drugs and being paid under
the current average sales price
Iv. Responding To President Trump’s Call To Action | 25 |
(ASP) methodology. The CAP,
or a model building on CAP
authority, may provide
opportunities for Federal savings
to the extent that aggregate bid
prices are less than 106 percent of
ASP, and provides opportunities
for physicians who do not wish
to bear the financial burdens and
risk associated with being in the
business of drug acquisition.
• Working in conjunction with the
Department of Commerce, the U.S.
Trade Representative, and the U.S.
Intellectual Property Enforcement
Coordinator to develop the
knowledge base necessary to
address the unfair disparity
between the drug prices in America
and other developed countries.
The Trump Administration
is committed to making the
appropriate regulatory changes
and seeking legislative solutions to
put American patients first.
C. Lowering List Prices
In response to President Trump’s
call to action, HHS may:
• Call on the FDA to evaluate the
inclusion of list prices in direct-
to-consumer advertising.
• Direct CMS to make Medicare and
Medicaid prices more transparent,
hold drug makers accountable for
their price increases, highlight
drugs that have not taken price
increases, and recognize when
competition is working with an
updated drug pricing dashboard.
This tool will also provide
patients, families, and caregivers
with additional information to
make informed decisions and
predict their cost sharing.
• Develop proposals related to the
Aordable Care Act’s Maximum
Rebate Amount provision, which
limits manufacturer rebates on
brand and generic drugs in the
Medicaid program to 100% of the
Average Manufacturer Price.
D. Reduce Patient
Out-of-Pocket Spending
In response to President Trump’s
call for action, HHS may:
• Prohibit Part D plan contracts
from preventing pharmacists
from telling patients when they
could pay less out-of-pocket by
not using their insurance – also
known as pharmacy gag clauses.
• Require Part D Plan sponsors to
provide additional information
about drug price increases and
lower-cost alternatives in the
Explanation of Benefits they
currently provide their members.
| 26 | American Patients First
V. Further Actions Under Review
and Opportunities for Feedback
BUILDING ON THE IDEAS already pro-
posed, HHS is considering even
bolder actions to bring down pric-
es for patients and taxpayers. These
include new measures to increase
transparency; fix the incentives that
may be increasing prices for pa-
tients; and reduce the costs of drug
development. HHS is interested in
public comments about how the de-
partment can take action to improve
competition and end the gaming of
regulatory processes, support bet-
ter negotiation of drug discounts
through government insurance pro-
grams, create incentives for phar-
maceutical companies to lower list
prices, and reduce consumer out-
of-pocket spending at the pharmacy
and other care settings. HHS is also
interested in public comments about
the general structure and function
of the pharmaceutical market, to
inform these actions. Proposals de-
scribed in this section are for admin-
istrative action, when within agency
authority, and legislative proposals
as necessary.
HHS is soliciting comments on
these and other policies under active
consideration.
A. Increasing competition
Underpricing or Cost-Shifting. Do
HHS programs contain the correct
incentives to obtain aordable pric-
es on safe and eective drugs? Does
the Best Price reporting requirement
of the Medicaid Drug Rebate Program
pose a barrier to price negotiation
and certain value-based agreements
in other markets, or otherwise shift
costs to other markets? Are govern-
ment programs causing underpricing
of generic drugs, and thereby reduc-
ing long-term generic competition?
Aordable Care Act taxes and re-
bates. The Aordable Care Act im-
posed tens of billions of dollars in
new taxes and costs on drugs sold in
government programs through a new
excise tax, an increase in the Medicaid
drug rebate amounts, and an exten-
sion of these higher rebates to com-
mercially-run Medicaid Managed
Care Organizations. How have these
changes impacted manufacturer
list pricing practices? Are govern-
ment programs being cross-subsi-
dized by higher list prices and excess
costs paid by individuals and em-
ployers in the commercial market?
If cross-subsidization exists, are the
taxes and artificially-depressed pric-
es causing higher overall drug costs
or other negative eects?
Access to reference product samples
Distribution restrictions. Certain
prescription drugs are subject to

V. Further Actions Under Review And Opportunities For Feedback | 27 |
limitations on distribution. Some
of these distribution limitations are
imposed by the manufacturer, while
others may be imposed in connec-
tion with an FDA-mandated Risk
Evaluation and Mitigation Strategy
(REMS). Some manufacturers may
be gaming these distribution lim-
itations to prevent generic devel-
opers from accessing their drugs
to conduct the tests that are legal-
ly required for a generic drug to be
brought to market, thereby limiting
opportunities for competition that
could place downward pressure on
drug prices. In some instances, for
products that are subject to REMS
that impact distribution, manufac-
turers continue to restrict access to
generic developers even after the
FDA issues a letter stating that it has
favorably evaluated the develop-
er’s proposed safety protections for
testing and would not consider the
provision of drug samples to this de-
veloper for generic development to
violate the applicable REMS. Should
additional steps be taken to review
existing REMS to determine whether
distribution restrictions are appro-
priate? Are there terms that could
be included in REMS, or provided in
addition to REMS, that could expand
access to products necessary for ge-
neric development? Are there other
steps that could be taken to facilitate
access to products that are under
distribution limitations imposed by
the manufacturer?
Samples for biosimilars and
interchangeables. Like some ge-
Some manufacturers
may be gaming
distribution
limitations to
prevent generic
developers
from accessing
their drugs.
neric drug developers, companies
engaged in biosimilar and inter-
changeable product development
may encounter diculties obtain-
ing sucient samples of the ref-
erence product for testing. What
actions should be considered to fa-
cilitate access to reference product
samples by these companies?
Biosimilar development, approval,
education, and access
Resources and tools from FDA: FDA
prioritizes ongoing eorts to im-
prove the eciency of the biosimilar
and interchangeable product devel-
opment and approval process. For
example, FDA is working to identify
areas in which additional informa-
tion resources or development tools
may facilitate the development of
high-quality biosimilar and inter-
| 28 | American Patients First
changeable products. What specif-
ic types of information resources or
development tools would be most
eective in reducing the develop-
ment costs for biosimilar and inter-
changeable products?
Improving the Purple Book. In
the Purple Book, FDA publishes in-
formation about biological prod-
ucts licensed under section 351 of the
Public Health Service Act, including
reference products, biosimilars, and
interchangeable products. The Purple
Book provides information about
these products that is useful to pre-
scribers, pharmacists, patients, and
other stakeholders. FDA is committed
to the timely publication of certain
information about reference prod-
uct exclusivity in the Purple Book.
How could the Purple Book be more
useful to health care professionals,
patients, manufacturers, and other
stakeholders? What additional infor-
mation could be added to increase the
utility of the Purple Book?
Educating providers and pa-
tients. Physician and patient con-
fidence in biosimilar and inter-
changeable products is critical to
the increased market acceptance of
these products. FDA intends to build
on the momentum of past educa-
tion eorts, such as the launch of its
Biosimilars Education and Outreach
Campaign in 2017, by developing
additional resources for health care
professionals and patients. What
types of information and educa-
tional resources on biosimilar and
interchangeable products would be
most useful to heath care profes-
sionals and patients to promote un-
derstanding of these products? What
role could state pharmacy practice
acts play in advancing the utilization
of biosimilar products?
Interchangeability. How could
the interchangeability of biosimilars
be improved, and what eects would
it have on the prescribing, dispens-
ing, and coverage of biosimilar and
interchangeable products?
B. Better Negotiation
The American pharmaceutical mar-
ketplace is built on innovation and
competition. However, regula-
tions governing how Medicare and
Medicaid pay for prescription drugs
have not kept pace with the avail-
ability of new types of drugs, partic-
ularly higher-cost curative therapies
intended for use by fewer patients.
Drug companies, commercial insur-
ers, and states have proposed cre-
ative approaches to financing these
new treatments, including indica-
tion-based pricing, outcomes-based
contracts, long-term financing
models, and others. Value-based
transformation of our entire health-
care system is a top HHS priority.
Improving price transparency is an
important part of achieving this aim.
What steps can be taken to improve
price transparency in Medicare,
Medicaid, and other forms of health
coverage, so that consumers can
seek value when choosing and using
their benefits?
V. Further Actions Under Review And Opportunities For Feedback | 29 |
Value-Based Arrangements and
Price Reporting. What benefits would
accrue to Medicare and Medicaid
beneficiaries by allowing manufac-
turers to exclude from statutory price
reporting programs discounts, re-
bates, or price guarantees included
in value-based arrangements? How
would excluding these approach-
es from Average Manufacturer Price
(AMP) and Best Price (BP) calcu-
lations impact the Medicaid Drug
Rebate program and supplemental
rebate revenue? How would these
exclusions aect Average Sales Price
(ASP) and 340B Ceiling Prices? What
benefits would accrue to Medicare
and Medicaid beneficiaries by ex-
tending the time for manufacturers
to report restatements of AMP and/
or BP reporting, as outlined in 42 CFR
§447.510, to accommodate adjust-
ments because of possible extended
Value-based Purchasing (VBP) eval-
uation timeframes? Is there a time-
frame CMS should consider that will
allow manufacturers to restate AMP
and BP without negative impact on
state rebate revenue? What modifi-
cations could be made to the follow-
ing regulatory definitions in the cur-
rent Medicaid Drug Rebate Program
that could facilitate the development
of VBP arrangements: 1) bundled
sale; 2) free good; 3) unit; or 4) best
price? Would providing specific AMP/
BP exclusions for VBP pricing used
for orphan drugs help manufactur-
ers that cannot adopt a bundled sale
approach? What regulatory changes
would Medicaid Managed Care or-
ganizations find helpful in negotiat-
ing VBP supplemental rebates with
manufacturers? How would these
changes aect Medicare or the 340B
program? Are there particular sec-
tions of the Social Security Act (e.g.,
the anti-kickback statute), or other
statutes and regulations that can be
revised to assist with manufacturers’
and states’ adoption of value-based
arrangements?
Indication-based payments. Pre-
scription drugs have varying degrees
of eectiveness when used to treat dif-
ferent types of disease. Though drugs
may be approved by the FDA to treat
specific indications, or used o-la-
bel by prescribers to treat others, they
are typically subject to the same price.
Should Medicare or Medicaid pay the
same price for a drug regardless of the
diagnosis for which it is being used?
How could indication-based pricing
support value-based purchasing?
What lessons could be learned from
private health plans? Are there unin-
tended consequences of current low-
cost drugs increasing in price due to
their identification as high value? How
and by whom should value be deter-
mined?? Is there enough granularity
in coding and reimbursement systems
to support indication-based pricing?
Are changes necessary to CMS’s price
reporting program definitions or how
the FDA’s National Drug Code num-
bers are used in CMS price reporting
programs? Do physicians, pharma-
cists, and insurers have access to all
the information they need to support
indication-based payments?
| 30 | American Patients First
Long-term fnancing models.
States and other payers typically es-
tablish budgets or premium rates for
a given benefit year. As such, their
budgets may be challenged when a
new high-cost drug unexpected-
ly becomes available in the benefit
year. Long-term financing models
are being proposed to help states,
insurers, and consumers pay for
high-cost treatments by spread-
ing payments over multiple years.
Should the state, insurer, drug man-
ufacturer, or other entity bear the
risk of receiving future payments?
How should Medicare or Medicaid
account for the cost of disease avert-
ed by a curative therapy paid for by
another payer? What regulations
should CMS consider revising to al-
low manufacturers and states more
flexibility to participate in novel
value-based pricing arrangements?
What eects would these solutions
have on manufacturer development
decisions? What current barriers
limit the applicability of these ar-
rangements in the private sector?
What assurances would parties need
to participate in more of these ar-
rangements, particularly with re-
gard to public programs?
Part B Competitive Acquisition
Program. HHS has the authority to
operate a Competitive Acquisition
Program for Part B drugs. What
changes would vendors and pro-
viders need to see relative to the
2007-2008 implementation of this
program in order to successful-
ly participate in the program? Has
the marketplace evolved such that
there would be more vendors capa-
ble of successfully participating in
this program? Are there a sucient
number of providers interested in
having a vendor selected through
a competitive bidding process ob-
tain these drugs on their behalf, and
bear the financial risk and carrying
costs? How could this program be
implemented in a way that ensures
a competitive market among multi-
ple vendors? Is it necessary that the
vendors also hold title to the drugs
and provide a distribution channel
or are there other ways they can pro-
vide value? What other approaches
could lower Part B drug spending for
patients of providers choosing not to
participate, without restricting their
access to care?
Part B to D. The President’s
Budget requested the authority to
move some Medicare Part B drugs
to Medicare Part D. Which drugs or
classes of drugs would be good candi-
dates for moving from Part B to Part
D? How could this proposal be imple-
mented to help reduce out-of-pock-
et costs for the 27% of beneficiaries
who do not have Medicare prescrip-
tion drug coverage, or those who
have Medicare supplemental benefits
in Part B? What additional informa-
tion would inform how this proposal
could be implemented and operated?
Part B drugs are reportedly avail-
able to OECD nations at lower prices
than those paid by Medicare Part B
providers. HHS is interested in re-
ceiving data describing the dier-

V. Further Actions Under Review And Opportunities For Feedback | 31 |
ences between the list prices and
net prices paid by Medicare Part B
providers, and the prices paid for
these same drugs by OECD nations.
Though these national health sys-
tems may be demanding lower pric-
es by restricting access or delaying
entry, should Part B drugs sold by
manufacturers oering lower prices
to OECD nations be subject to nego-
tiation by Part D plans? Would this
lead to lower out-of-pocket costs on
behalf of people with Medicare? How
could this aect access to medicines
for people with Medicare?
Fixing Global Freeloading. U.S.
consumers and taxpayers general-
ly pay more for brand drugs than do
consumers and taxpayers in oth-
er OECD countries, which often have
reimbursements set by their central
government. In eect, other countries
are not paying an appropriate share of
the necessary research and develop-
ment to bring innovative drugs to the
market and are instead freeriding o
U.S. consumers and taxpayers. What
can be done to reduce the pricing dis-
parity and spread the burden for in-
centivizing new drug development
more equally between the U.S. and
other developed countries? What pol-
icies should the U.S. government pur-
sue in order to protect IP rights and
address concerns around compulsory
licensing in this area.
Site neutrality for physician-ad-
ministered drugs. Currently un-
der Medicare Part B and often in
Medicaid, hospitals and physi-
cians are reimbursed comparable
“We want to look at
negotiating for drugs
in Medicare that
have never been
negotiated for.”
– SECRETARY AZAR
amounts for drugs they adminis-
ter to patients, but the facility fees
when drugs are administered at
hospitals and hospital-owned out-
patient departments are many times
higher than the fees charged by phy-
sician oces. What eect would a
site neutral payment policy for drug
administration procedures have on
the location of the practice of med-
icine? How would this change aect
the organization of health care sys-
tems? How would this change aect
competition for health care services,
particularly for cancer care?
Site neutrality between inpatient
and outpatient setting. Medicare
payment rules pay for prescription
drugs dierently when provided
during inpatient care (Part A) or ad-
ministered by an outpatient physi-
cian (Part B). Beneficiaries also have
dierent cost-sharing requirements
in Part A and Part B. Some drugs can
be administered in either the in-
patient or outpatient setting, while

| 32 | American Patients First
“For high-cost
products, the
amount that
Medicare pays
diers dramatically
based on the
site of service.”
– CMS ADMINISTRATOR
SEEMA VERMA
others are currently limited to in-
patient use because of safety con-
cerns. Do the dierences between
Medicare’s Part A and Part B drug
payment policies create aordabil-
ity and access challenges for bene-
ficiaries? What policies should CMS
consider to ensure inpatient and
outpatient providers are neither
underpaid nor overpaid for a drug,
regardless of where it was admin-
istered? Which elements of the in-
patient or outpatient setting lead to
naturally dierential payments, and
why? If a drug can be used safely in
the outpatient setting, and achieve
the same outcomes at a lower cost,
how should Medicare encourage the
shift to outpatient settings? In what
instances would inpatient adminis-
tration actually be less costly?
Accuracy of national spending
data. Are annual reports of health
spending obscuring the true cost of
prescription drugs? What is the val-
ue of better understanding the dif-
ference between gross and net drug
prices? How could the Medicare
Trustees Report, annual National
Health Expenditure publications,
Uniform Rate Review Template, and
other publications more accurately
collect and report gross and net drug
spending in medical and pharmacy
benefits? Should average Part D re-
bate amounts be reported separately
for small molecule drugs, biologics,
and high-cost drugs? What inno-
vation is needed to maximize price
transparency without disclosing
proprietary information or data pro-
tected by confidentiality provisions?
C. Create Incentives
to Lower List Prices
Government programs, commercial
insurers, and individual consumers
pay for drugs dierently. The price
paid at the pharmacy counter or re-
imbursed to a physician or hospital
is the result of many dierent com-
plex financial transactions between
drug makers, distributors, insur-
ers, Pharmacy Benefits Managers,
pharmacies and others. Public pro-
grams are also subject to state and
federal regulations governing what
drugs are covered, who can be paid
for them, and how much will be paid.
Too often, these negotiations do not
result in the lowest out-of-pocket

V. Further Actions Under Review And Opportunities For Feedback | 33 |
costs for consumers, and may actu-
ally be causing higher list prices.
Fiduciary duty for Pharmacy
Benefit Managers. Pharmacy Benefit
Managers (PBMs) and benefits con-
sultants help buyers (insurers, large
employers) seek rebates intended to
lower net drug prices, and help sellers
(drug manufacturers) pay rebates to
secure placement on health plan for-
mularies. Most current PBM contracts
may allow them to retain a percentage
of the rebate collected and other ad-
ministrative or service fees.
Do PBM rebates and fees based on
the percentage of the list price cre-
ate an incentive to favor higher list
prices (and the potential for higher
rebates) rather than lower prices?
Do higher rebates encourage bene-
fits consultants who represent pay-
ers to focus on high rebates instead
of low net cost? Do payers manage
formularies favoring benefit designs
that yield higher rebates rather than
lower net drug costs? How are ben-
eficiaries negatively impacted by
incentives across the benefits land-
scape (manufacturer, wholesaler,
retailer, PBM, consultants, and in-
surers) that favor higher list prices?
How can these incentives be reset to
prioritize lower out-of-pocket costs
for consumers, better adherence and
improved outcomes for patients?
What data would support or refute
the premise described above?
Should PBMs be obligated to act
solely in the interest of the entity for
whom they are managing pharma-
ceutical benefits? Should PBMs be
Too oen,
negotiations do not
result in the lowest
out-of-pocket costs
for consumers.
forbidden from receiving any pay-
ment or remuneration from manu-
facturers, and should PBM contracts
be forbidden from including rebates
or fees calculated as a percentage of
list prices? What eect would im-
posing this fiduciary duty on PBMs
on behalf of the ultimate payer (i.e.,
consumers) have on PBMs’ ability to
negotiate drug prices? How could this
aect manufacturer pricing behav-
ior, insurance, and benefit design?
What unintended consequences for
beneficiary out-of-pocket spending
and federal health program spend-
ing could result from these changes?
Reducing the impact of rebates.
Increasingly higher rebates in fed-
eral health care programs may be
causing higher list prices in public
programs, and increasing the prices
paid by consumers, employers, and
commercial insurers. What should
CMS consider doing to restrict or
reduce the use of rebates? Should
Medicare Part D prohibit the use of
rebates in contracts between Part D
plan sponsors and drug manufac-

| 34 | American Patients First
Increasingly
higher rebates
in federal health
care programs
may be causing
higher list prices.
turers, and require these contracts
to be based only on a fixed price for
a drug over the contract term? What
incentives or regulatory chang-
es (e.g., removing the discount safe
harbor) could restrict the use of re-
bates and reduce the eect of rebates
on list prices? How would this aect
the behavior of drug manufacturers,
PBMs, and insurers? How could it
change formulary design, premium
rates, or the overall structure of the
Part D benefit?
Incentives to lower or not in-
crease list prices. Should manufac-
turers of drugs that have increased
their prices over a particular look-
back period or have not provided a
discount be allowed to be includ-
ed in the protected classes? Should
drugs for which a price increase has
not been observed over a particu-
lar lookback period be treated dif-
ferently when determining the ex-
ceptions criteria for protected class
drugs? What should CMS consider
doing, under current authorities,
to create incentives for Part D drug
manufacturers committing to a
price over a particular lookback pe-
riod? How long should the lookback
period be?
The Healthcare Common Pro-
cedure Coding System (HCPCS)
codes for new Part B drugs are not
typically assigned until after they
are commercially available. Should
they be available immediately at
launch for new drugs from man-
ufacturers committing to a price
over a particular lookback period?
What should CMS consider doing,
under current authorities, to create
incentives for Part B drugs com-
mitting to a price over a particular
lookback period? How long should
the lookback period be?
How could these incentives aect
the behavior of manufacturers and
purchasers? What are the operation-
al concerns to implementing them?
Are there other incentives that could
be created to reward manufacturers
of drugs that have not taken a price
increase during a particular look-
back period?
Infationary rebate limits. The
Department is concerned that lim-
iting manufacturer rebates on brand
and generic drugs in the Medicaid
program to 100% of calculated AMP
allows for excessive price increas-
es to be taken without manufactur-
ers facing the full eect of the price
inflationary penalty established by
Congress. This policy, implemented
as part of the ACA, may allow for run-
V. Further Actions Under Review And Opportunities For Feedback | 35 |
away price increases and cost-shift-
ing. When is this limitation a valid
constraint upon the rebates manu-
facturers should pay? What impacts
would removing the cap on the in-
flationary rebate have on list prices,
price increases over time, and public
and private payers?
Exclusion of certain payments,
rebates, or discounts from the deter-
mination of Average Manufacturer
Price and Best Price. The Department
is concerned that excluding pharma-
cy benefit manager rebates from the
determination of Best Price, imple-
mented as part of the ACA, may al-
low for runaway price increases and
cost-shifting. The Department is also
interested in learning more about the
eect of excluding payments received
from, and rebates or discounts pro-
vided to Pharmacy Benefit Managers
from the determination of Average
Manufacturer Price.
What impacts would these chang-
es have on list prices, price increas-
es over time, and public and private
payers? What data would support or
refute the premise described above?
Copay discount cards. Does the
use of manufacturer copay cards
help lower consumer cost or actu-
ally drive increases in manufactur-
er list price? Does the use of copay
cards incent manufacturers and
PBMs to work together in driving
up list prices by limiting the trans-
parency of the true cost of the drug
to the beneficiary? What data would
support or refute the premise de-
scribed above?
CMS regulations presently ex-
clude manufacturer sponsored drug
discount card programs from the de-
termination of average manufacturer
price and the determination of best
price. What eect would eliminating
this exclusion have on drug prices?
Would there be circumstances
under which allowing beneficiaries of
federal health care programs to uti-
lize copay discount cards would ad-
vance public health benefits such as
medication adherence, and outweigh
the eects on list price and concerns
about program integrity? What data
would support or refute this?
The 340B drug discount program
The 340B Drug Pricing Program was
established by Congress in 1992, and
requires drug manufacturers partic-
ipating in the Medicaid Drug Rebate
Program to provide covered outpa-
tient drugs to eligible health care
providers—also known as covered
entities—at reduced prices. Covered
entities include certain qualify-
ing hospitals and federal grantees
identified in section 340B of the
Public Health Service Act (PHSA).
The Health Resources and Services
Administration (HRSA) adminis-
ters and oversees the 340B Program,
and the discounts provided may af-
fect the prices paid for drugs used
by Medicare beneficiaries, people
with Medicaid, and those covered by
commercial insurance.
Program growth. The 340B
program has grown significantly
since 1992—not only in the num-

| 36 | American Patients First
Does the use of
manufacturer
copay cards help
lower consumer
cost or actually
drive increases
in manufacturer
list price?
ber of covered entities and contract
pharmacies, but also in the amount
of money saved by covered entities.
HRSA estimates that covered enti-
ties saved approximately $6 billion
on approximately $12 billion in dis-
counted purchases in Calendar Year
(CY) 2015 by participating in the
340B Program.
ii
It is estimated that
discounted drug purchases made by
covered entities under the 340B pro-
gram totaled more than $16 billion
in 2016—a more than 30 percent in-
crease in 340B Program purchases in
just one year.
iii
How has the growth
of the 340B drug discount program
aected list prices? Has it caused
cross-subsidization by increasing list
prices applicable in the commercial
sector? What impact has this had on
insurers and payers, including Part
D plans? Does the Group Purchasing
Organization (GPO) exclusion, the
establishment of the Prime Vendor
Program, and the current inventory
models for tracking 340B drugs in-
crease or decrease prices? What are
the unintended consequences of this
program? Would explicit general reg-
ulatory authority over all elements of
the 340B Program materially aect
the elements of the program aect-
ing drug pricing?
Program Eligibility. Would
changing the definition of “patient”
or changing the requirements gov-
erning covered entities contracting
with pharmacies or registering o-
site outpatient facilities (i.e., child
sites) help refocus the program to-
ward its intended purpose?
Duplicate Discounts. The 340B
statute prohibits duplicate dis-
counts. Manufacturers are not re-
quired to provide a discounted
340B price and a Medicaid drug re-
bate for the same drug. Are the cur-
rent mechanisms for identifying
and preventing duplicate discounts
effective? Are drug companies pay-
ing additional rebates over the
ii. 340B Drug Pricing Program Ceiling Price and Manufacturer Civil Monetary Penalties Regulation, 82 Fed. Reg.
1210, 1227 (Jan. 5, 2017).
iii. Aaron Vandervelde and Eleanor Blalock, Measuring the Relative Size of the 340B Program: 2012-2017, BERKE-
LEY RESEARCH GROUP (July 2017), available at
https://www.thinkbrg.com/media/publication/928_Vandervelde_Measuring340Bsize-July-2017_WEB_FINAL.pdf.

V. Further Actions Under Review And Opportunities For Feedback | 37 |
statutory 340B discounts for drugs
that have been dispensed to 340B
patients covered by commercial
insurance? What is the impact on
drug pricing given that private in-
surers oftentimes pay commercial
rates for drugs purchased at 340B
discounts? Do insurers, pharmacy,
PBM, or manufacturer contracts
consider, address, or otherwise
include language regarding drugs
purchased at 340B discounts? What
should be considered to improve
the management and the integri-
ty of claims for drugs provided to
340B patients in the overall insured
market? What additional oversight
or claims standards are necessary
to prevent duplicate discounts in
Medicaid and other programs?
Reduce Patient
Out-of-Pocket Spending
Part D end-of-year statement on
drug price changes and rebates col-
lected. Part D plans presently pro-
vide their members with an expla-
nation of benefits, which includes
information about the negotiated
price for each of their dispensed pre-
scriptions, and what the plan, mem-
ber, and others paid. What addition-
al information could be added about
the rate of change in those prices
over the course of the benefit year?
Alternatively, could pharmacists be
empowered to inform beneficiaries
when prices for their drugs have
changed? Would this information be
best distributed by pharmacists at
the point of sale, by Medicare as an
Discounted drug
purchases made
under the 340B
drug program
totaled more than
$16 billion in 2016.
annual report, or by the health plan
on a more regular basis, or some
combination of these approaches?
Could CMS improve transparency
for Medicare beneficiaries without
violating the Part D program’s con-
fidentiality protections? What oper-
ational challenges or concerns about
burden exist with this approach, and
how could CMS measure compliance
with this approach?
Federal preemption of con-
tracted pharmacy gag clause laws.
Right now, some contracts between
health plans and pharmacies do
not allow the pharmacy to inform
a patient that the same drug or a
competitor could be purchased at
a lower price o-insurance. What
purpose do these clauses serve oth-
er than to require beneficiaries pay
higher out-of-pocket costs? What
other communication barriers are
in place between pharmacists and
patients that could be impeding
lower drug prices, out-of-pocket
| 38 | American Patients First
costs, and spending? Should phar-
macists be required to ask patients
in federal programs if they’d like
information about lower-cost al-
ternatives? What other strategies
might be most eective in provid-
ing price information to consumers
at the point of sale?
Inform Medicare beneficiaries
with Medicare Part B and Part D
about cost-sharing and lower-cost
alternatives. Health plans and phar-
macy benefit managers have found
new ways to inform prescribers and
pharmacists, when prescribing or
dispensing a new prescription, about
the formulary options, expected
cost-sharing, and lower-cost alter-
natives specific to individual patients.
How could these tools reduce out-
of-pocket spending for people with
Medicare? Is this technology present
in all or most electronic prescribing
or pharmacy dispensing systems?
Should Medicare require the use of
systems that support providing this
information to patients? What exist-
ing systems, tools, or third-party ap-
plications could support the creation
of these tools? Does the technology
exist for this approach to be quick-
ly and inexpensively implement-
ed? Would this increase costs for the
Medicare program? Does this create
unreasonable burden for prescribers
or pharmacists?
V. Further Actions Under Review And Opportunities For Feedback | 39 |
Endnotes
1. Malkin, J. D., D. P. Goldman, and G. F. Joyce. “The Changing Face Of Pharmacy Benefit
Design.” Health Aairs 23, no. 1 (January/February 2004): 194-99.
2. “2016 Medicine Use and Spending in the U.S.,” IQVIA
3. PL 111-148; Payment for covered outpatient drugs, § 42 U.S. Code § 1396r–8
4. “Medicaid Managed Care Enrollment Reports, 2009 & 2010.” Medicaid.gov. Accessed April
23, 2018. https://www.medicaid.gov/medicaid/managed-care/enrollment/index.html.
5. “Overview of the 340B Drug Pricing Program.” MedPAC.gov. May 2015.
6. U.S. Congress. House. Energy & Commerce Committee. Review of the 340B Drug Pricing
Program. 115th Cong. H. Rept.
7. Fein, Adam J. “The 340B Program Hits $16.2 Billion in 2016; Now 5% of U.S. Drug Market.”
Drug Channels. May 18, 2017. Accessed April 23, 2018. http://www.drugchannels.net/2017/05/
exclusive-340b-program-hits-162-billion.html.
8. Rémuzat, C., D. Urbinati, J. Roïz, A. Kornfeld, and M. Toumi. “Overview Of External Reference
Pricing Systems In Europe.” Journal of Market Access & Health Policy 17, no. 3 (September 15, 2015).
9. “External Price Referencing.” WHO Collaborating Centre for Pharmaceutical Pricing and
Reimbursement Policies. http://whocc.goeg.at/Glossary/PreferredTerms/External price
referencing.
10. https://www.k.org/report-section/ehbs-2017-section-9-prescription-drug-benefits/;
https://www.cdc.gov/nchs/data/nhis/earlyrelease/ERHDHP Access 0617.pdf.
11. Shrank, William H., Niteesh K. Choudhry, Michael A. Fischer, Jerry Avorn, Mark Powell,
Sebastian Schneeweiss, Joshua N. Liberman, Timothy Dollear, Troyen A. Brennan, and M.
Alan Brookhart. “The Epidemiology of Prescriptions Abandoned at the Pharmacy.” Annals of
Internal Medicine 153, no. 10 (November 16, 2010): 633.
12. IQVIA proprietary data.


