
GHTF/SG3/N18:2010
FINAL DOCUMENT
Global Harmonization Task Force
Title: Quality management system –Medical Devices – Guidance on corrective
action and preventive action and related QMS processes
Authoring Group: Study Group 3
Date: 4 November 2010
Dr. Larry Kelly, GHTF Chair
The document herein was produced by the Global Harmonization Task Force, which is comprised of representatives
from medical device regulatory agencies and the regulated industry. The document is intended to provide non-
binding guidance for use in the regulation of medical devices, and has been subject to consultation throughout its
development.
There are no restrictions on the reproduction, distribution or use of this document; however, incorporation of this
document, in part or in whole, into any other document, or its translation into languages other than English, does not
convey or represent an endorsement of any kind by the Global Harmonization Task Force.
Copyright © 2010 by the Global Harmonization Task Force

Guidance on corrective action and preventive action and related QMS processes GHTF/SG3/N18:2010
November 4, 2010 Page 2 of 26
Table of Contents
Preface ........................................................................................................................................................................3
Introduction.................................................................................................................................................................3
1.0 Scope...............................................................................................................................................................4
2.0 Definitions.......................................................................................................................................................4
2.1 Correction .........................................................................................................................................4
2.2 Corrective action...............................................................................................................................4
2.3 Data Sources .....................................................................................................................................4
2.4 Concession........................................................................................................................................4
2.5 Preventive action...............................................................................................................................5
2.6 Nonconformity..................................................................................................................................5
2.7 Verification .......................................................................................................................................5
2.8 Validation..........................................................................................................................................5
3.0 Overview.........................................................................................................................................................5
4.0 Phase I: Planning.............................................................................................................................................7
4.1 Plan for Measurement, Analysis and Improvement Processes..........................................................8
4.2 Establish Data Sources and Criteria..................................................................................................9
5.0 Phase II: Measurement and Analysis within and across Data Sources..........................................................10
5.1 Measure...........................................................................................................................................10
5.2 Analyze...........................................................................................................................................10
6.0 Phase III: Improvement.................................................................................................................................14
6.1 Investigate.......................................................................................................................................14
6.2 Identify Root Cause ........................................................................................................................16
6.3 Identify Actions ..............................................................................................................................17
6.4 Verify Identified Actions ................................................................................................................18
6.5 Implement Actions..........................................................................................................................18
6.6 Determine Effectiveness of Implemented Actions..........................................................................19
7.0 Phase IV: Input to Management....................................................................................................................19
7.1 Report to Management....................................................................................................................19
7.2 Management Review.......................................................................................................................20
Annex A: Examples of Phase Activities...................................................................................................................21
Annex B: Examples of Data Sources and Data Elements.........................................................................................22
Annex C: Examples of Contributing Factors............................................................................................................24
Annex D: Examples for Documentation of the Improvement Processes..................................................................25

Guidance on corrective action and preventive action and related QMS processes GHTF/SG3/N18:2010
November 4, 2010 Page 3 of 26
Preface
The document herein was produced by the Global Harmonization Task Force, a voluntary group
of representatives from medical device regulatory agencies and the regulated industry. The doc-
ument is intended to provide non-binding guidance for use in the regulation of medical devices,
and has been subject to consultation throughout its development.
There are no restrictions on the reproduction, distribution or use of this document; however, in-
corporation of this document, in part or in whole, into any other document, or its translation into
languages other than English, does not convey or represent an endorsement of any kind by the
Global Harmonization Task Force.
Introduction
This guidance document is intended for medical device manufacturers and regulatory authorities.
It is intended for educational purposes and is not intended to be used to assess or audit compli-
ance with regulatory requirements. It is expected that the reader is familiar with regulatory Qual-
ity Management System (QMS) requirements within the medical devices sector.
For the purposes of this document it is assumed that the medical device manufacturer has a QMS
which requires the manufacturer to have documented processes to ensure that medical devices
placed on the market are safe and effective. For example ISO13485 Medical Devices – Quality
Management Systems – Requirements for regulatory purposes, Japanese Ministerial Ordinance
on Standards for Manufacturing Control and Quality Control for Medical Devices and in vitro
Diagnostics (MHLW
1
Ministerial Ordinance No. 169), the FDA
2
Quality System Regulation 21
CFR Part 820 or the respective quality system requirements of the European medical Device Di-
rectives.
For this purpose the manufacturer will establish processes and define appropriate controls for
measurement and analysis to identify nonconformities and potential nonconformities. Also, the
manufacturer should establish processes defining when and how corrections, corrective actions,
or preventive actions should be undertaken. These actions should be commensurate with the sig-
nificance or risk of the nonconformity or potential nonconformity.
The terms risk, risk management and related terminology utilized within this document are in
accordance with ISO 14971 “Medical Devices-Application of Risk Management to Medical De-
vices.”
The acronym “CAPA” will not be used in this document because the concept of corrective action
and preventive action has been incorrectly interpreted to assume that a preventive action is re-
quired for every corrective action.
1
Japanese Ministry of Health Labor and Welfare
2
US Food and Drug Administration

Guidance on corrective action and preventive action and related QMS processes GHTF/SG3/N18:2010
November 4, 2010 Page 4 of 26
This document will discuss the escalation process from different “reactive” sources which will
be corrective in nature and other “proactive” sources which will be preventive in nature. The
manufacturer is required to account for both types of data sources whether they are of a correc-
tive or preventive nature.
Regardless of the nature of the data source, if there is a decision to escalate the information to
further evaluation and investigation, the steps of investigation, identification of root causes and
actions needed, verification, implementation, and effectiveness checks will be similar.
This guidance document will describe measurement, analysis and improvement as complete and
integrated processes.
1.0 Scope
This document provides guidance for establishing adequate processes for measurement, analysis
and improvement within the QMS as related to correction and/or corrective action for noncon-
formities or preventive action for potential nonconformities of systems, processes or products.
2.0 Definitions
The references to clauses in this section refer to ISO 9000:2005.
2.1 Correction
Action to eliminate a detected nonconformity (3.6.2)
Note 1 A correction can be made in conjunction with corrective action (3.6.5)
Note 2 Corrections can be, for example, rework (3.6.7) or re-grade (3.6.8)
2.2 Corrective action
Action to eliminate the cause of a detected nonconformity (3.6.2) or other undesirable situ-
ation
Note 1 There can be more than one cause for nonconformity
Note 2 Corrective action is taken to prevent recurrence whereas preventive ac-
tion (3.6.4) is taken to prevent occurrence
Note 3 There is a distinction between correction (3.6.6) and corrective action
2.3 Data Sources
The processes within a Quality Management System that provide quality information that
could be used to identify nonconformities, or potential nonconformities
2.4 Concession
Permission to use or release a product that does not conform to specified requirements
(3.6.11).

Guidance on corrective action and preventive action and related QMS processes GHTF/SG3/N18:2010
November 4, 2010 Page 5 of 26
2.5 Preventive action
Action to eliminate the cause of a potential nonconformity (3.6.2) or other undesirable sit-
uation
Note 1 There can be more than one cause for nonconformity
Note 2 Preventive action is taken to prevent occurrence whereas corrective ac-
tion (3.6.5) is taken to prevent recurrence
2.6 Nonconformity
Non fulfillment of a requirement (3.1.2)
2.7 Verification
Confirmation through provision of objective evidence (3.8.1) that specified requirements
(3.1.2) have been fulfilled
Note 1 The term “verified” is used to designate the corresponding status.
Note 2 Confirmation can comprise activities such as:
- performing alternative calculations,
- comparing a new design specification (3.7.3) with a similar proven
design specification, undertaking tests (3.8.3), performing demonstra-
tions, and reviewing and approving documents prior to issue.
2.8 Validation
Confirmation through provision of objective evidence (3.8.1) that the requirements for a
specific intended use or application have been fulfilled
Note 1 The term “validated” is used to designate the corresponding status.
Note 2 The use conditions for validation can be real or simulated.
3.0 Overview
The manufacturer is responsible for the implementation and maintenance of a QMS which en-
ables their organization to provide safe and effective medical devices meeting customer and
regulatory requirements.
A nonconformity as defined in 2.6 is a non fulfillment of a requirement. It is important to under-
stand that requirements may relate to product, process or the QMS.
When a nonconformity is identified, the manufacturer will determine the significance, the asso-
ciated risk and the potential for recurrence.
Once these have been determined the manufacturer may decide the nonconformity has little as-
sociated risk or is unlikely to recur. In such cases the manufacturer may decide only to carry out
a correction.

Guidance on corrective action and preventive action and related QMS processes GHTF/SG3/N18:2010
November 4, 2010 Page 6 of 26
Should the nonconformity recur within the QMS, during manufacture or after the medical device
has been delivered to a customer, it is an indication that improvement action(s) may be needed.
In either case the QMS requires that a corrective action should be carried out with the aim to
prevent recurrence. The corrective action may be as simple as retraining, or as complex as redes-
igning the manufacturing process.
The manufacturer may encounter situations that have not actually caused a nonconformity, but
may do so in the future. Such situations may call for preventive action. For example, production
or acceptance testing trend data indicates that control limits are being approached and revision of
product or production (process, equipment or facilities) requirements may be necessary. These
revisions could constitute a preventive action. Preventive action would not include planned
process adjustments intended to return process performance to nominal values from the edges of
the process control range.
Actions taken to eliminate observed nonconformities within the scope of a single QMS (regard-
less of whether the actions are taken at more than one site or facility operating within that QMS)
would be considered corrective actions. However, similar actions applied within another QMS
(regardless of whether it is the same site, facility, or organization) that has not yet experienced
these nonconformities, would be considered preventive actions.
Figure 1 illustrates typical Phases to be considered when planning, implementing and maintain-
ing effective processes for measurement, analysis, improvement and providing input to manage-
ment. See Annex A for a list of possible activities corresponding to the phases in Figure 1.
As a check on the effectiveness of the processes defined, management should regularly review
the outputs of processes and make adjustments as needed.
Documented procedures, requirements and records should be maintained by the manufacturer to
ensure and demonstrate the effective planning, operation and control of the processes. Docu-
mented evidence of decisions and actions taken will be a part of the QMS.

Guidance on corrective action and preventive action and related QMS processes GHTF/SG3/N18:2010
November 4, 2010 Page 7 of 26
Figure 1: Processes for measurement, analysis and improvement
4.0 Phase I: Planning
Planning involves specifying processes and associated resources in order to meet specific objec-
tives. Factors to consider during the planning phase should be aligned with the manufacturer’s
overall business planning and include the device’s intended use, markets and users, as well as
regulatory requirements.
The involvement of management at appropriate levels (e.g. review, approval) in actions taken in
response to nonconformities or potential nonconformities should be established. Management
should ensure that measurement criteria are defined for identified data sources and communi-
cated across the organization.

Guidance on corrective action and preventive action and related QMS processes GHTF/SG3/N18:2010
November 4, 2010 Page 8 of 26
4.1 Plan for Measurement, Analysis and Improvement Processes
Factors to consider during this planning phase should be aligned with the manufacturer’s overall
business planning and as a minimum include the type of device being manufactured, intended
markets and users, and regulatory requirements. As part of planning, management should review
the processes critical to the operations with regard to quality and regulatory requirements and
select relevant data sources to measure, analyze and facilitate improvement as necessary.
In the process of planning measurement and analysis, a manufacturer needs to take into account
data sources, the measurement of the data elements within each data source, the frequency of
monitoring, and the analysis to be performed within a data source, or across data sources.
The measurement of data elements should be done in a way that ensures the manufacturer is ef-
fective in managing the operations and maintain an effective QMS. Each of the data elements
should be planned and established with specific requirements for measurement that are moni-
tored routinely.
The scope of the QMS and the scope of the measurement, analysis and improvement processes
will provide the boundaries as to whether the data source is reactive/corrective or proac-
tive/preventive.
The planning phase should ensure the following:
Identification of relevant internal and external data sources that are indicators of process
and product performance.
Provision for adequate resources and establish responsibilities and authorities to enable
the necessary actions. Resources may include technical experts, testing laboratories, data
management, infrastructure, training, etc.
Definition of requirements for each identified data source, including limits, acceptance
criteria, escalation criteria and mechanisms for reporting of nonconformities or potential
nonconformities.
Analysis of data elements within data sources.
Coordination and analysis of data across data sources.
For each data element individual criteria should be defined; however, criteria may be defined for
a combination of data elements. Criteria should be quantitative whenever possible in order to
maximize consistency and reproducibility for subsequent analysis. If the criteria and data are qu-
alitative, subjectivity should be eliminated or minimized.
Acceptance criteria should be based on system, product and process specifications or require-
ments which are typically identified during design and development activities. This includes the
design of the Quality Management System, development and maintenance of assembly proc-
esses, delivery processes, servicing and installation processes.
Escalation criteria used for the purpose of initiating the improvement process (see 6.0) may often
be called action levels, trigger points, thresholds, etc. These escalation criteria should be proce-
duralized and would likely include certain generic action levels as well as specific action levels
resulting from risk management activities. In particular, criteria should be established for imme-

Guidance on corrective action and preventive action and related QMS processes GHTF/SG3/N18:2010
November 4, 2010 Page 9 of 26
diate escalation. For example, an incident alleging a death or serious injury should be escalated
to the improvement phase (see 6.0) for immediate action.
For new technology and existing technologies with new intended uses/applications, initial escala-
tion criteria may be difficult to define for the monitoring process. Therefore a manufacturer
should plan for resources to analyze information in order to confirm initial assumptions and es-
tablish or revise escalation criteria.
Planning should provide for confirmation that the defined limits, acceptance criteria, escalation
criteria and mechanisms for reporting of nonconformities or potential nonconformities for the
original data sources and data elements are still appropriate. Where new data sources need to be
established, confirm that they have been identified and their criteria defined.
4.2 Establish Data Sources and Criteria
The manufacturer should identify and document relevant data sources and their data elements,
both internal and external to the organization. Data elements provide information regarding non-
conformities, potential nonconformities and the effectiveness of the established processes within
the data sources.
Examples of data sources can be, but are not restricted to:
Regulatory Requirements
Management Review
Supplier (performance/controls)
Complaint Handling
Adverse Event Reporting
Process Controls
Finished Product
Quality Audits (internal/external)
Product Recall
Spare Parts Usage
Service Reports
Returned Product
Market/Customer Surveys
Scientific Literature
Media Sources
Product Realization (design, purchasing, production and service and customer informa-
tion)
Risk Management
For further examples of data elements see Annex B.
When an issue is identified in one of the data sources, it is also important that the manufacturer
identify and review related information from other data sources across the organization. Fur-
thermore a review of information from external data sources should also be considered. The ag-
gregation of information from more than the original data source may lead to more comprehen-

Guidance on corrective action and preventive action and related QMS processes GHTF/SG3/N18:2010
November 4, 2010 Page 10 of 26
sive knowledge. With this knowledge base a manufacturer will be positioned to better determine
appropriate action.
5.0 Phase II: Measurement and Analysis within and across Data Sources
Once data sources, data elements and acceptance criteria have been specified, as part of the plan-
ning process, the manufacturer is required to perform measurement, monitoring and analysis
processes to determine conformity or nonconformity.
Software used in measurement, monitoring and analysis, whether purchased (Off-The-Shelf) or
custom developed, should be validated for its intended use.
For example, a customer survey conducted by the marketing department, indicated that there was
a general dissatisfaction with the packaging of product X. When investigated further (within and
across other data sources) and reviewed with other data from complaints, returned product and
service reports, it became evident that there was a potential for misuse, unsafe use, or damage to
the device as a result of the current packaging design. As the result of this analysis, escalation to
Phase III (see 6.0) for preventive action may be appropriate.
5.1 Measure
For the purpose of this guidance, measurement is a set of operations to determine a value of a
data element (i.e. quantity, quality).
Data collected from the measurement of product, process and QMS are acquired throughout the
life-cycle of the product. The manufacturer should define for example frequency of the meas-
urement, precision and accuracy of the data. The manufacturer should also ensure that the data
collected is current and relevant.
Measurement data should be retained as a quality record. The manufacturer should maintain the
data in a form that is retrievable, suitable for analysis and meets both QMS and regulatory re-
quirements.
Monitoring is the systematic and regular collection of a measurement. The manufacturer should
define during the planning phase what, when and how data should be monitored. The data should
be defined such that it can be analyzed for further action. The monitoring of data may be con-
tinuous or periodic, depending on the type of data source and elements. The monitoring proc-
esses should be periodically reviewed for their continued suitability.
5.2 Analyze
For the purpose of this guidance, analysis is a systematic review and evaluation of data from
measurements to derive a conclusion.

Guidance on corrective action and preventive action and related QMS processes GHTF/SG3/N18:2010
November 4, 2010 Page 11 of 26
The manufacturer should have documented procedures for the analysis of data against the estab-
lished criteria (see 4.1). Analysis is performed to identify nonconformity or potential noncon-
formity or identify areas where further investigation should be initiated. In addition analysis is
used to demonstrate the suitability and effectiveness of product, process and QMS. Analysis can
be performed utilizing analytical tools, a team of experts, process owners or independent review-
ers. The results of the analysis should be documented.
After it is determined what will be measured, statistical techniques should be identified to help
understand variability and thereby help the manufacturer to maintain or improve effectiveness
and efficiency. These techniques also facilitate better use of available data to assist in decision
making. Statistical techniques assist in identifying, measuring, analyzing, interpreting and mod-
eling variability.
For the analysis of nonconformity, appropriate statistical and non-statistical techniques can be
applied. Examples for statistical techniques are:
Statistical Process Control (SPC) charts
Pareto analysis
Data trending
Linear and non-linear regression analysis
Experimental design (DOE – Design of Experiments) and analysis of variance
Graphical methods (histograms, scatter plots, etc.)
Non-statistical techniques are for example:
Management reviews
Results from quality meetings
Safety committees (internal/external)
Failure Mode and Effect Analysis (FMEA)
Fault Tree Analysis (FTA)
Analysis will likely occur at several different points (time and/or organizational level). For ex-
ample, a certain amount of analysis and possible failure investigation (where there is evidence of
a nonconformity) will occur for each data source.
In addition to the analysis within the data sources there should also be a level of analysis across
data sources to determine the extent and significance of nonconformity or potential nonconform-
ity. The linkage of data from different data sources will be referred to as “horizontal analysis”.
The horizontal analysis may:
determine that the action proposed from the data source analysis is appropriate without
further progress into Phase III (see 6.0); or,
provide additional information warranting progress into Phase III (see 6.0), regardless of
whether the data source analysis escalated the nonconformity or potential nonconformity.
The outcome of measurement and analysis leads to different scenarios as shown in Figure 2.
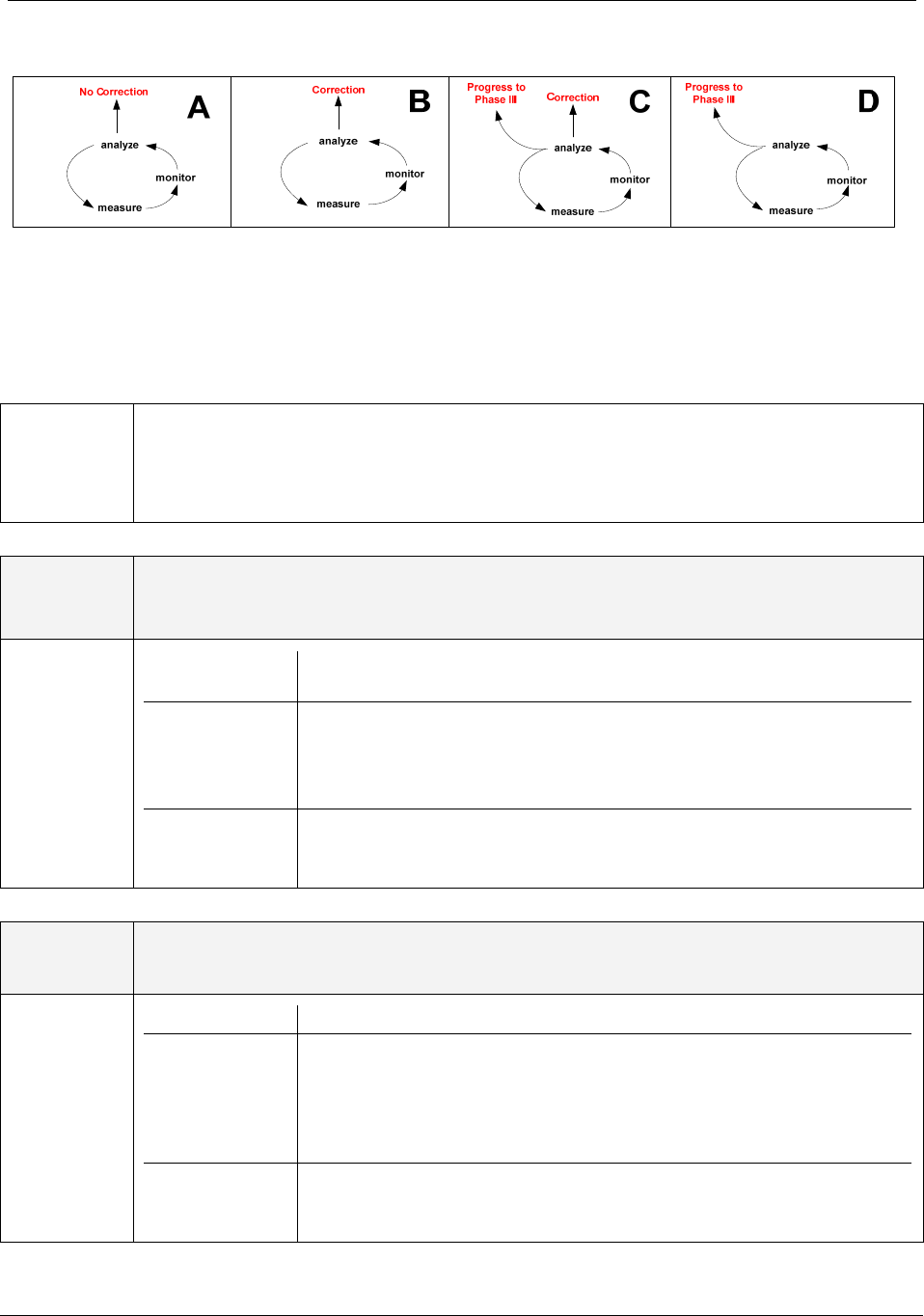
Guidance on corrective action and preventive action and related QMS processes GHTF/SG3/N18:2010
November 4, 2010 Page 12 of 26
Figure 2: Outcomes of measurement and analysis
The following tables provide more details to support the use of Figure 2. Each scenario is de-
scribed with an example showing the different outcome of measurement and analysis.
Basic
Example
The documentation requirements in a research design and development procedure
were not followed. The missing documentation involves changing to a different
supplier of an electronic board. The requirement is to document the supplier name
and supplier number in the research report.
Scenario A
No correction required, continue measurement and monitoring
The decision is made not to take any correction nor escalate the handling of the nonconformity to
Phase III (see 6.0).
Example
Nonconformity The supplier number was not included in the research report. (however, the
supplier name is documented).
Key Results of
Measurement
and Analysis
Analysis indicates that the procedure is adequate and well known to the us-
ers of the research procedure.
Following a review of the issue this appears to be a one time oversight.
The intent of the requirement is for convenience only.
Conclusion No initial correction - It is not necessary to update the research report, as the
supplier is documented by name, hence traceability is maintained.
Do not escalate to Phase III.
Scenario B
Correction required, continue measurement and monitoring
The decision is made to perform a correction but not to escalate the handling of the nonconformity
to Phase III (see 6.0).
Example
Nonconformity The supplier name and number was not included in the research report.
Key Results of
Measurement
and Analysis
Analysis indicates that the procedure is adequate and well known to the us-
ers of the research procedure.
Following a review of the issue this appears to be a one time oversight.
The intent of the requirement is to ensure traceability to the supplier and this
could be lost if the research report is not updated.
Conclusion Take an initial correction to update the research report with the supplier
name and number.
Do not escalate to Phase III.

Guidance on corrective action and preventive action and related QMS processes GHTF/SG3/N18:2010
November 4, 2010 Page 13 of 26
Scenario C
Correction and escalation to further investigation under the improvement phase.
The decision is made to perform an initial correction. However, there is a need for escalation to
Phase III (see 6.0) to further investigate as a result of the analysis performed in order to determine
the appropriate corrective action.
Example
Nonconformity The supplier name and number was not included in the research report.
Key Results of
Measurement
and Analysis
Analysis indicates that the procedure may not be adequate and it is not well
know to the users of the research procedure. The issue has been identified in
multiple reports.
In some cases, traceability to the supplier could be established via other
means, and in other cases it could not.
Conclusion Take an initial correction to update the research report with the supplier
name and number (in the cases where the supplier could be identified).
Escalate to Phase III for corrective action.
Scenario D
Escalation for further investigation under the improvement phase.
The decision is made that there is not enough information at this time to determine the required
action. Therefore the investigation is escalated to Phase III.
Example
Nonconformity The supplier name and number was not included in the research report.
Key Results of
Measurement
and Analysis
Analysis indicates that the procedure may not be adequate and it is not well
know to the users of the research procedure. The issue has been identified in
multiple reports.
Traceability to the supplier could not be established via other means in any
of the cases.
Conclusion No initial correction - The supplier is not known so an initial correction can-
not be taken at this time.
Escalate to Phase III for corrective action.
Documented procedures should clearly delineate and define when escalation to Phase III is re-
quired.
Typically manufacturers have organizational groups or processes surrounding some of their main
data sources (e.g. complaint handling, handling of nonconformities, material review boards,
change management process). Within these groups or processes certain activities described in
Figure 2 can be implemented without escalation.
There may be predefined events that due to the significance of the risk will be escalated to Phase
III without any delay that can not be justified. In the event a potential nonconformity is identi-
fied, it may be escalated into Phase III (see 6.0) for consideration of actions to prevent the occur-
rence of the potential nonconformity.
When no correction or only corrections within these groups or processes are taken, there needs to
be data source monitoring and analysis (e.g. trending) to determine if escalation to Phase III may
be necessary from accumulated information. Whenever an issue is escalated to Phase III, any in-
formation gained within the defined activities of these groups or processes should be an input to
the Phase III activities such as Investigation (see 6.1) or Identified Actions (see 6.3).

Guidance on corrective action and preventive action and related QMS processes GHTF/SG3/N18:2010
November 4, 2010 Page 14 of 26
6.0 Phase III: Improvement
The improvement phase of a corrective action process or preventive action process is designed to
eliminate or mitigate a nonconformity or potential nonconformity.
The improvement activities are dependant on the specific nonconformity or potential noncon-
formity. Any previous data from Phase II should be utilized as input to the Phase III process.
The improvement phase and the activities described in Figure 3 needs to be documented. Im-
provement generally involves the following activities that the manufacturer would take sequen-
tially or sometimes simultaneously:
A thorough investigation of the reported nonconformity
An in-depth root cause analysis
Identification of appropriate actions
Verification of identified actions
Implementation of actions
Effectiveness check of implemented actions
Improvement
6.1 Investigate
6.2 Identify Root Cause
6.3 Identify Actions
6.4 Verification of
identified Actions
6.5 Implement Actions
6.6 Determine
Effectiveness of
Implemented Actions
Figure 3: Phase III – Improvement
6.1 Investigate
The purpose of investigation is to determine the root cause of existing or potential non –
conformities, whenever possible, and to provide recommendations of solutions. The magni-
tude/scope of the investigation should be commensurate with the determined risk of the noncon-
formity.
Good practice shows that a documented plan should be in place prior to conducting the investi-
gation (see Annex D for examples). The plan should include:
Description of the nonconformity expressed as a problem statement
Scope of the investigation
Investigation team and their responsibilities
Description of activities to be performed
Resources
Methods and tools
Timeframe

Guidance on corrective action and preventive action and related QMS processes GHTF/SG3/N18:2010
November 4, 2010 Page 15 of 26
From the information obtained throughout the process the problem statement should be reviewed
and refined as appropriate.
The investigation should:
Determine the extent of the nonconformity or potential nonconformity
Acknowledge that there are likely to be several causes of an event; hence, the investiga-
tion should not cease prematurely
Require that symptoms be distinguished from root causes and advocate the treatment of
root causes rather than just the symptoms
Require that an end point be defined for the investigation. An overly exhaustive investi-
gation may unduly delay the correction of non-conformity or unnecessarily incur addi-
tional cost. (For example, if removal of the causes identified so far will correct 80% of
the effects then it is likely that the significant causes have been identified (Pareto rule))
Take into account the output of relevant risk management activities
Agree on the form of evidence. For example, evidence should support:
- the seriousness of the event
- the likelihood of occurrence of the event
- the significance of the consequences flowing from the event
The investigation should include the collection of data to facilitate analysis and should build
upon any analysis, evaluation and investigation that were previously performed (see 5.0). This
will require the investigator to identify, define and further document the observed effects/non-
conformity, or already determined causes, to ensure that the investigator understands the context
and extent of the investigation. It may be necessary to:
Review and clarify the information provided
Review any additional information available from an horizontal analysis
Consider whether this is a systemic issue/non-systemic issue
Gather additional evidence, if required
Interview process owners/operators or other parties involved
Review documents
Inspect facilities, or the environment of the event
Previous investigations should be reviewed in order to determine if the event is a new problem or
the recurrence of a previous problem where, for example, an ineffective solution was imple-
mented. The following questions will assist in making the determination:
Is the nonconformity from a single data source?
Does the current nonconformity correlate with nonconformities from other data sources?
Are multiple data sources identifying the same nonconformity?
Do other nonconformities have an effect on the problem investigated here?
Many of the tools used in investigations rely upon a cause and effect relationship between an
event and a symptom of that event. To ensure that causes are identified, not symptoms, the fol-
lowing should be considered:
There must be a clear description of a cause and its effect. The link between the cause
and the undesirable outcome needs to be described.
Each description of a cause must also describe the combined conditions that contribute to
the undesired effect

Guidance on corrective action and preventive action and related QMS processes GHTF/SG3/N18:2010
November 4, 2010 Page 16 of 26
A failure to act is only considered a cause if there was a pre-existing requirement to act. The re-
quirement to act may arise from a procedure, or may also arise from regulations, standards or
guidelines for practice, or other reasonably expected actions.
Some of the more common tools and techniques include:
Cause and effect diagrams
5 Why’s analysis
Pareto charting
Fishbone/Ishikawa cause and effect diagrams
Change analysis
Risk analysis techniques
Is/Is Not
The outcome of an investigation should include:
Clearly defined problem statement
What information was gathered, reviewed and/or evaluated
Results of the reviews/evaluations of the information
Identification of cause(s) or contributing factors
Solutions to address the cause(s) or contributing factor(s)
6.2 Identify Root Cause
Causes or contributing factors of detected nonconformity or potential nonconformity should
promptly be identified so that corrective action can be taken to prevent recurrence, or preventive
action taken to prevent occurrence. The process to identify the root cause should start with the
output(s) of the investigation (see 6.1).
When assessing relevant data, the following should be considered:
Systematic generation of cause and effect conclusions supported by documented evidence
Evaluate significant or underlying causes and their relationship to the problem
Ensure that causes are identified, not the symptoms
Check for more than one root cause (above processes if necessary)
Causes or contributing factors of nonconformities or potential nonconformities may include the
following:
Failure of, or malfunction of, incoming materials, processes, tools, equipment or facilities
in which products are processed, stored or handled, including the equipment and systems
therein
Inadequate or non-existent procedures and documentation
Non-compliance with procedures
Inadequate process control
Inadequate scheduling
Lack of training
Inadequate working conditions
Inadequate resources (human or material)
(Inherent) process variability

Guidance on corrective action and preventive action and related QMS processes GHTF/SG3/N18:2010
November 4, 2010 Page 17 of 26
For further details on aspects to be considered when doing the root cause analysis see Annex C.
The output of the root cause analysis should be a clear statement of the most fundamental
cause(s) resulting in the nonconformity (see Annex D for examples).
6.3 Identify Actions
When the root cause(s) has been determined, the manufacturer should identify and document the
necessary corrections and/or corrective actions or preventive actions. These actions should be
reviewed to ensure that all necessary actions are identified. The review may benefit from a cross
functional approach. Where applicable, product disposition decisions should also be docu-
mented.
The following outcomes are possible and should be documented:
No further action necessary
(provided that no safety issue exists and regulatory requirements are met)
- With continuous monitoring
- Acceptance under concession and continuance of monitoring
Correction
It may be necessary to take initial corrections (e.g. containment, stop of shipment/supply,
issuance of advisory notice) in order to address an immediate risk or safety issue. This
may be necessary before investigation has been completed and root cause has been de-
termined. However, after investigation and root cause determination, additional and/or
possibly different corrections may become necessary.
Corrective action
Corrective action should address systemic problems. For example, changing the proce-
dure and training of personnel to the revised procedure may not, by itself, be appropriate
or sufficient to address the systemic cause(s).
Preventive action
By its very nature preventive action can not follow a nonconformity.
As a result of this step, a list of action items should be documented. These may include:
A detailed description of the implementation
Review regulatory requirements (e.g. submissions, licensing, certifications)
Roles and responsibilities for execution of action items
Identification of the necessary resources (e.g. IT, infrastructure, work environment)
Verification and/or validation protocols of the action(s) with acceptance criteria
Implementation schedule, including timelines
Method or data for the determination of effectiveness with acceptance criteria
Identify the starting point of monitoring, and end point of correction and/or corrective ac-
tion or preventive action as described above

Guidance on corrective action and preventive action and related QMS processes GHTF/SG3/N18:2010
November 4, 2010 Page 18 of 26
6.4 Verify Identified Actions
Before the implementation of action(s), a manufacturer should verify the identified action(s) and
approve their implementation. In addition validation may be required where process validation or
re-validation may be necessary, or where user needs or intended uses are changed and design va-
lidation will be required.
Verification activities are to ensure that all the elements of the proposed action (documentation,
training etc) will satisfy the requirements of the proposed action. These activities should be per-
formed by persons who are knowledgeable in the design or use of the product or process that is
the subject of corrective or preventive action. Verification of a preventive action can be accom-
plished by introducing the conditions that would induce a nonconformity and confirming that the
nonconformity does not occur.
Validation activities generate data and information that confirm the likelihood of the effective-
ness of the corrective action to eliminate the nonconformity or proposed nonconformity.
Examples of items to be considered when planning the verification/validation activities include:
Does the action(s) eliminate the identified root cause(s)?
Does the action(s) cover all affected products/processes?
Does the action(s) adversely affect the final products?
Is it possible to finalize the actions timely in planned schedule
(resources, materials/kits, logistics, communications, etc.)?
Is the execution of the action commensurate with the degree
of risk previously established?
Are new risks or nonconformities derived from the action?
6.5 Implement Actions
The following items that may be considered at implementation should be documented:
Parties involved
Materials
Processes
Training
Communications
Tools
Timelines for the implementation of the approved action
Verify that the implementation has been completed.

Guidance on corrective action and preventive action and related QMS processes GHTF/SG3/N18:2010
November 4, 2010 Page 19 of 26
6.6 Determine Effectiveness of Implemented Actions
The manufacturer should gather data over a period of time related to the effectiveness of the im-
plemented action (see Annex D for examples).
Management should ensure and be involved in a review and confirmation that actions taken were
effective and did not introduce new issues or concerns. The following questions should be con-
sidered at appropriate times throughout the process and be revisited in the final review:
Has the problem been comprehensively identified?
Has the extent of the problem been identified (e.g. range of affected devices, patient out-
come, process, production lines, operator)?
Have the root cause/contributing factors of the problem been identified and addressed?
Has the improvement action(s) been defined, planned, documented, verified and imple-
mented?
If the manufacturer finds the actions are not effective, the manufacturer should re-initiate Phase
III activities (see 6.0). If the manufacturer finds the actions create a new issue or a new noncon-
formity then the manufacturer needs to initiate Phase II (see 5.0) activities.
7.0 Phase IV: Input to Management
Management at different levels in the organization should be involved in each improvement ac-
tion either through approval of the improvement steps or reporting.
The Management Review is the overall mechanism for management to ensure that the Quality
Management System as a whole is effective.
7.1 Report to Management
The manufacturer should have a mechanism/procedure that expeditiously raises safety related
issues or other high risk issues to management. These issues can be identified in the data sources,
the improvement phase (see 6.0), or originate from other sources external to the Quality Man-
agement System. In addition to this expeditious escalation mechanism, the manufacturer should
define management and personnel responsibilities (i.e. process owner) for the measurement,
analysis and improvement processes, to ensure that the processes and the actions being imple-
mented are effective. For this purpose there needs to be a mechanism for management at differ-
ent levels to stay informed of the information or data from:
The measurement and analysis activities from the individual data sources
The investigations, actions, implementations, etc. from the improvement processes

Guidance on corrective action and preventive action and related QMS processes GHTF/SG3/N18:2010
November 4, 2010 Page 20 of 26
7.2 Management Review
The manufacturer has procedures for what is provided as input for the management review, in-
cluding relevant information from the improvement processes, such as improvement actions
(corrective actions, and/or preventive actions) as well as important corrections.
The manufacturer needs to define what meaningful data is to be reported for a management re-
view. Data should be specific to the quality objectives of the manufacturer and be reported regu-
larly. Merely providing the number of improvement actions or the number of how many im-
provement actions are opened or closed to the management review process are not sufficient in
assessing the effectiveness of the processes.
Included in this review would be an assessment of any opportunities for improvement of the de-
vice, manufacturing process, QMS or the organization itself.
An outcome of the review could be the allocation of funding or personnel to a particular area,
project or device that the review has identified as not meeting customer and regulatory safety and
effectiveness expectations.

Guidance on corrective action and preventive action and related QMS processes GHTF/SG3/N18:2010
November 4, 2010 Page 21 of 26
Annex A: Examples of Phase Activities
List of possible activities corresponding to the phases in Figure 1.
The following is an outline/aid memoir of the main points described in this document. It is not
intended as a “box ticking” exercise and should not be used as such, but used purely to summa-
rize and align the steps in the process described in this document. The activity numbers do not
imply sequential steps – some steps may take place in parallel.
The references in this Annex refer to the sections in this document.
Phase Activities
Planning
1. Identify all data sources (internal/external) by product type (4.1)
2. Identify resources required and individual personnel responsibilities for
measuring each data source (4.1)
3. Define the requirements for each data source and the data elements
within each data source that will be measured and analysed (4.1)
4. Define requirements for escalation to the improvement phase (4.1)
5. Define requirements for monitoring the measurements in the data
sources (5.1)
6. Establish data sources (4.2)
Measurement and Analysis
within and across Data Sources
7. Measure and analyse all data sources for nonconformities and potential
nonconformities (5.0, 5.1 and 5.2)
8. Have reports of nonconformity or potential nonconformity come from
more than one data source?
9. Is the nonconformity or potential nonconformity systemic?
Improvement 10. Determine scope and required outcome of investigation (6.1)
11. Investigate nonconformity or potential nonconformity (6.1)
12. Analyse nonconformity or potential nonconformity for root cause(s)
(6.2)
13. Identify actions ( correction, corrective action or preventive action)
(6.3)
14. Verify proposed actions before implementation (6.4)
15. Implement proposed actions (6.5)
16. Determine effectiveness of actions (validate if possible) (6.6)
Input to Management 17. Report investigation and outcome to management (7.1)
18. Review investigation, analysis and outcome (6.6, 7.2)
19. If not satisfied return to step 10
20. If required, report to regulator (note: reporting may be required earlier
depending on severity)*
21. Audit system at determined intervals*
22. If numbers of nonconformities or potential nonconformities exceeds
targets, review all QMS processes*
*
Steps 20 to 22 are not described in this document but are added as reminders of general management responsibilities
in this area of the QMS.

Guidance on corrective action and preventive action and related QMS processes GHTF/SG3/N18:2010
November 4, 2010 Page 22 of 26
Annex B: Examples of Data Sources and Data Elements
Examples of data sources and their data elements can be, but are not restricted to:
Data Sources Data Elements
Regulatory Requirements Result of a regulatory inspection
New or revised regulatory requirements
Management Review Management review output
Supplier
Performance/Controls
Number of batches received
Batch and/or shipment
Inspection and test records
Quantity of rejects or deviations
Reason for rejection
By supplier, if more than one supplier
Use in which product or service
Supplier problems
Complaint Handling
Quantity
By product family
By customer (physician, healthcare facility, patient, etc.)
Reason for complaint
Complaint codes
Severity
Component involved
Adverse Event Reporting Event
Quantity
By product family
By customer (physician, healthcare facility, patient, etc.)
Type of event (death or serious injury, etc.)
Component involved
Process Controls By product
Operator
Work shift
Equipment and/or instruments used
Inspection and test records
In-process control results
Process control parameters
Inspection process
Final acceptance
Rejects
Special process
Validation study results
Process monitoring observations
Finished Product Inspection and test records
Quality Audits
(internal/external)
Observations (number, category, corporate policy, regulatory requirements,
significance, etc.)
Repeat observations (indicative of effectiveness)
Closure times
Overall acceptability of contractor or supplier
Compliance to audit schedule
Audit personnel

Guidance on corrective action and preventive action and related QMS processes GHTF/SG3/N18:2010
November 4, 2010 Page 23 of 26
Data Sources Data Elements
Product Recall Timeliness of recall communication
Classification of recall
Recall effectiveness checks
Spare Parts Usage
Frequency of replacement
Batch number of spare part
By supplier of spare part, if more than one supplier
By customer
By location or area of customer
Service Reports Installation
First use of equipment
Frequency of maintenance visits
Types of repairs
Frequency of repairs
Usage frequency
Parts replaced
Service personnel
Returned Product Quantity
Reason for returning product
By customer
Types of defects identified on returned product
Market/Customer Sur-
veys
Customer preferences
Customer service response time
Solicited information on new or modified products
Scientific Literature Research papers
Media Sources Articles in trade journals
Product Realization
( Design, Purchasing,
Production and Service
and Customer informa-
tion)
Design and development review results
Design and development verification results
Design and development validation results
Design and development changes
(reason or cause for change, effectiveness of change, etc.)
Controls on purchased products or services
(See above Supplier Performance/Controls)
Verification results of purchased product
Inspection and testing data of purchased product
Production and Service processes-
Cleaning operations of product and facilities
Sterilization
Installation results
Servicing and Maintenance if required (See also: Service Reports)
Verification and Validation results of processes used in production and
service. Including approval of equipment and qualification of personnel
Traceability Data
Controls of monitoring and measuring devices
Calibration and maintenance of equipment
Customer Information- New or repeat customer
Customer feedback maybe in other forms than complaints or returned
product (Customer Service call data, repeat sales , delivery/distribution data)
Risk Management Published reports/literature of failures of similar products
Stakeholder concerns and generally accepted state of the art
Risk acceptability criteria

Guidance on corrective action and preventive action and related QMS processes GHTF/SG3/N18:2010
November 4, 2010 Page 24 of 26
Annex C: Examples of Contributing Factors
Examples of possible contributing factors to be considered when doing the root cause analysis:
Materials
Defective raw material (does material meet specification?)
Batch related problem
Design problem (wrong material for product, wrong specifications)
Supplier problem (lack of control at supplier, alternative supplier)
Lack of raw material.
Machine / Equipment
Incorrect tool selection – suitability
Inadequate maintenance or design – calibration?
Equipment used as intended by the manufacturer?
Defective equipment or tool
End of life?
Human error – inadequate training?
Environment
Orderly workplace
Properly controlled – temperature, humidity, pressure, cleanliness
Job design/layout of work
Management
Inadequate management involvement
Stress demands
Human factors
Hazards not properly guarded
Were management informed / did they take action?
Methods
Procedures not adequately defined
Practice does not follow prescribed methodology
Poor communications
Management system
Training or education lacking
Poor employee involvement
Poor recognition of hazard
Previous hazards not eliminated
Measurement, monitoring and improvement
Inadequate measuring and improvement

Guidance on corrective action and preventive action and related QMS processes GHTF/SG3/N18:2010
November 4, 2010 Page 25 of 26
Annex D: Examples for Documentation of the Improvement Processes
The table below includes guidance for documenting various requirements of the improvement
processes.
Guidance Example Documentation
Problem
Statement
Clearly defined problem statement.
State how the issue was discov-
ered. The process/procedure that
was not followed.
Provide evidence
What, When, Who, Where and
How much (as applicable)
During in-process testing of Product A finished product on
[date], two devices out of 30 were found to be noncon-
forming per Design Document 123456, revision A. Note
2.1 in Design Document 123456 requires that the surface
finish be 32 µinch maximum on all exterior surfaces. The
two nonconforming devices had a surface finish above the
maximum 32 µinch finish as follows:
Serial Number 54321 had a surface finish of 67 µinch
Serial Number 65432 had a surface finish of up to 38
µinch
Correction General Examples
Containment,
Stop of shipment/ supply
Issuance of advisory notice
Incident awareness / training
Change or suspend production
process
The supplier was notified of the issue on [date].
The supplier conducted an operator awareness training of
the incident on [date].
Initial extent of the issue is restricted to supplier lot #678.
All unused components and product built with components
from this lot were controlled on [date]. No product built
with this lot had been distributed.
Investigate
• Clearly defined problem statement
(update/refine if new information
is determined)
What information was gathered,
reviewed and/or evaluated
Results of the reviews/evaluations
of the information
Identification of cause(s) or con-
tributing factors
See initial problem statement. Subsequent investigation
confirmed that the issue was limited to lot #678. All addi-
tional available lots of this component were inspected with
a 95/95 inspection plan and no additional lots were con-
firmed to have the issue.
The incoming inspection process and component FMEA
were reviewed and determined to be adequate and accu-
rate, respectively.
Review of finished product reject data over the past year
revealed no other rejects for surface finish of this compo-
nent.
The following problem-solving tools and methods were
used during the course of the investigation of the surface
finish issue.
Fishbone analysis – see the attached file labeled ‘Sur-
face Finish Analysis’.
Conference calls and documentation reviews with the
Supplier – see attached file which contains the min-
utes from the conference calls.
Results of the investigation were the following. Two dif-
ferent raw tubing lots were mixed at the Supplier’s finish-
ing process. One raw tubing lot was intended for customer
A’s products (Lot number 10000-100 requiring a surface
finish of 32 µinch maximum) and the other was intended
for customer B’s product which had a surface finish above
the 32 µinch maximum.

Guidance on corrective action and preventive action and related QMS processes GHTF/SG3/N18:2010
November 4, 2010 Page 26 of 26
Identify
Root Cause
The output of the root cause analy-
sis should be a clear statement of
the most fundamental cause(s) re-
sulting in the nonconformity
It has been concluded that the root cause of the tubing sur-
face finish issue is inadequate line clearance procedures
established at the supplier.
Planned
actions
Specify:
What the action is
Who will do it
When it should be done
Corrective action: Supplier to add line clearance require-
ments to documented procedures by [date].
Preventive action: Not applicable.
Verification
of actions
Verification activities are to ensure
that all the elements of the pro-
posed action (documentation,
training etc) will satisfy the re-
quirements of the proposed action
Validation activities generate data
and information that confirm the
likelihood of the effectiveness of
the corrective action to eliminate
the nonconformity or proposed
nonconformity.
General examples are included below. Actual documenta-
tion would need to be more specific.
Review and approval of the procedural changes prior
to use
Conduct a pilot of new procedure on a specific pro-
ject/department/time frame prior to full scale imple-
mentation
Verification that the updated supplier procedure ad-
dresses the process that caused the nonconformity
Verification that the training materials address the
specific process that caused the nonconformity
Comparing a new design specification with a similar
proven design specification
Performing calculations using an alternative method
Perform validation of equipment, software, production
processes, test method, component, etc.
Specific example:
Review and approval of supplier procedure XXX by the
supplier and the customer to ensure adequacy of the up-
dated line clearance process.
Verification
of effective-
ness
Method or data for the determination of
effectiveness with acceptance criteria.
The improvement goal
The evidence (data sources) that
will be used to support effective-
ness (e.g., a data source could be
where the problem was initially
found)
The time frame that effectiveness
will be monitored (e.g., upon com-
pletion of actions or three months,
six months as appropriate)
OR
Sample size required to demon-
strate effectiveness
X months after implementation:
Conduct a query of the electronic manufacturing data
system to verify there are zero surface finish rejects
for this component at finished Product A final inspec-
tion.
Supplier Quality Engineer to conduct on site review at
the supplier of the action to confirm the procedures
are in place, are known to the operators, and there is
evidence that the procedures are being followed.
Winterhufen 1.0
